Advertisements
Advertisements
Question
Write reactions for the following conversions.
4-Nitrobenzoic acid to Nitrobenzene
Solution
Sodium salts of 4-nitrobenzoic acid on heating with soda lime give nitrobenzene.

APPEARS IN
RELATED QUESTIONS
Write the preparation reactions for acid amide from the following.
Carboxylic acid
Write reactions for the following conversions.
Propanone to Propane
Write chemical reactions to convert –COOH group of acetic acid into the following.
CH4
Write chemical reactions to convert –COOH group of acetic acid into the following.
C2H5OH
Write chemical reactions to convert –COOH group of acetic acid into the following.
CH3COCl
Write reaction for preparation of acetophenone from benzoyl chloride.
In the reaction,
\[\ce{CH3COOH ->[SOCl2] X ->[Sodium salt of carboxylic acid] Y}\].
The compound Y was found to be a mixed acid anhydride. Thus, the sodium salt of carboxylic acid used CANNOT be ____________.
Which is the gas evolved when carboxylic acids react with strongly electropositive metals (such as Na, K, Ca, Zn)?
The product of the reaction between dialkyl cadmium and acyl chloride is a/an ____________.
In the following reaction:
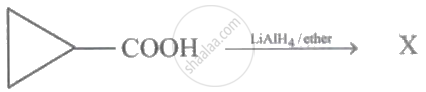
The product X is:
Products in the following reaction are:
\[\ce{CH3COOH + PCl3 ->[\Delta]}\]
What amount of dinitrogen contains 3.6 × 1018 molecules?
The reducing agent preferred to convert carboxylic acids and esters to primary alcohols is ____________.
The oxidation number of B in NaBH4 is ____________.
Which of the following aromatic acids has less acidic strength than benzoic acid?
Which of the following carboxylic acids ha highest acidic strength?
Which oxide of nitrogen is obtained on heating ammonium nitrate at 250°C?
Excess of ammonia with sodium hypochloride solution in the presence of glue or gelatine gives ______.
The number of bonding pairs and lone pairs of electrons present in the central atom of ammonia molecular are respectively.
Which of the following can reduce?
\[\ce{RCOOH -> RCH2OH}\]
How sodium bicarbonate test is used to distinguish between carboxylic acid and phenol?
How will you prepare Acetic anhydride from acetic acid?
