SSC (English Medium)
SSC (Marathi Semi-English)
Academic Year: 2024-2025
Date: March 2025
Advertisements
General Instructions :
- All questions are compulsory.
- Use of a calculator is not allowed.
- The numbers to the right of the questions indicate fu ll marks.
- In case of MCQs (Q. No. 1(A)) only the first attempt will be evaluated and will be given credit.
- For each MCQ, the correct alternative (A), (B), (C), or (D) with a subquestion number is to be written as an answer.
For Eg. : (i) (A), (ii) (B), (iii) (C) - Scientifically correct, labelled diagrams should be drawn wherever necessary.
______ react with dil. HNO3 to evolve hydrogen gas.
Iron and copper
Manganese and magnesium
Zinc and manganese
Aluminium and magnesium
Chapter: [0.09] Carbon Compounds
What happens when a light ray passes from a rarer medium to a denser medium?
It bends towards the normal.
It bends away from the normal.
It does not change its direction and hence does not refracted.
None of these
Chapter: [0.06] Refraction of Light [0.17] Wonders of Light 2
Alkaline earth metals have valency 2. This means that their position in the modern periodic table is in ______.
Group 2
Group 16
Period 2
d-block
Chapter: [0.02] Periodic Classification of Elements
In a double displacement reaction ______.
Ions remain a state.
Ions get liberated.
Ions are exchanged.
Ions are not created.
Chapter: [0.03] Chemical Reactions and Equations [0.12] The Magic of Chemical Reactions
Which of the following statement is incorrect for ionic compounds?
Ionic compounds are crystalline in nature.
Melting points of ionic compounds are low.
Ionic compounds are hard and brittle.
Ionic compounds have electrostatic attraction between them.
Chapter:
Find the odd one out:
Conduction
Convection
Radiation
Expansion
Chapter: [0.05] Heat
Complete the correlation:
Farsightedness : Convex lens : : Presbyopia : ______
Chapter: [0.07] Lenses
State true or false.
Bauxite is the main ore of aluminium.
True
False
Chapter: [0.08] Metallurgy
Match the columns:
| Column ‘A’ | Column ‘B’ |
| The density of water is maximum at | (a) 0°C |
| (b) 4°C | |
| (c) 100°C |
Chapter:
Write the IUPAC name of acetaldehyde CH3 – CHO.
Chapter:
Advertisements
Give a scientific reason.
Tungsten metal is used to make a solenoid type coil in an electric bulb.
Chapter: [0.04] Effects of Electric Current [0.14] The Electric Spark
It is recommended to use air tight containers for storing oil for a long time.
Chapter: [0.03] Chemical Reactions and Equations [0.12] The Magic of Chemical Reactions
Give scientific reasons.
Any object on earth falls vertically downward and not at an angle to the vertical; nor fly off in a horizontal direction.
Chapter: [0.01] Gravitation
Answer the following questions based on the given graph.

- Identify which curve shows AC and DC.
- What is the frequency of AC and DC?
Chapter: [0.04] Effects of Electric Current
If in a medium, the speed of light is 1.5 × 108 m/s how much will the absolute refractive index of that medium be?
Chapter: [0.06] Refraction of Light [0.17] Wonders of Light 2
Solve the problem.
If the mass of a planet is eight times the mass of the Earth and its radius is twice the radius of the Earth, what will be the escape velocity for that planet?
Chapter: [0.01] Gravitation
Complete the following chemical reaction:
\[\ce{CuSO4 (aq) + Pb (s) -> \underline{}\underline{}\underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}\underline{}\underline{}}\]
Name the chemical reaction type.
Chapter: [0.03] Chemical Reactions and Equations [0.12] The Magic of Chemical Reactions
Observe the given figure of reactivity series of metals and answer the following questions:

Reactivity series of metals
- Name two metals which react with water.
- Name two moderately reactive metals.
- Name the most highly reactive metal and the most less reactive metal.
Chapter: [0.08] Metallurgy
Advertisements
Explain the following temperature vs time graph.

Chapter: [0.05] Heat
The reaction between acetic acid and sodium carbonate is shown in the following figure.
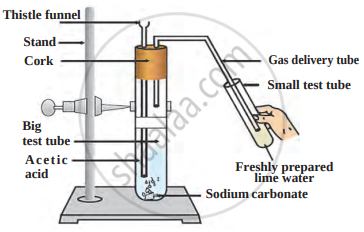
Answer the questions with the help of a diagram.
- Which gas does come out as effervescence in the big test tube?
- What is the colour change in the lime water present in the small test tube?
- Write the related reaction.
Chapter: [0.09] Carbon Compounds
| A process of forming a thick oxide of aluminium when aluminium is exposed to air. This coat makes it resistant to corrosion. Resistance can be improved by making a layer of oxide thinker. In this technique, the aluminium article is the anode, and the electrolyte is sulphuric acid. The anode reaction results in the formation of a black-coloured film of aluminium oxide on the anode. By putting appropriate dyes in the electrolytic solution, both coloured surface with the decorative finish is achieved. Kitchen articles like anodised such as pressure cookers, pans and frames of sliding windows are applications of this technique. |
- Name the anode and electrolyte used in this technique.
- How can we make aluminium articles made resistant to corrosion?
- Name the technique used to coat the aluminium articles.
Chapter: [0.03] Chemical Reactions and Equations [0.08] Metallurgy
Tara, after completing her exams, went to her grandmother's house along with her younger brother Rahul. One day, Rahul came home after playing and switched on the fan and air conditioner. At the same time, his grandmother was preparing a milkshake for him using a mixer. Suddenly, they heard a big sound and the electricity of the house got shut down. Tara is called an electrician. He said the main reason for supply to shut down was overloading.
- What is overloading?
- Is overloading and short the same? When does short-circuiting take place?
- How can the effects of overloading be avoided?
Chapter: [0.04] Effects of Electric Current [0.14] The Electric Spark
Observe the following diagram and answer the questions.

- Explain the construction of the equipment shown in the figure.
- How is AC generated?
Chapter: [0.15] All about Electromagnetism
An element has its electron configuration as 2, 8, 2. Now answer the following question.
What is the atomic number of this element?
Chapter: [0.02] Periodic Classification of Elements
An element has its electron configuration as 2, 8, 2. Now answer the following question.
What is the group of this element?
Chapter: [0.02] Periodic Classification of Elements
An element has its electron configuration as 2, 8, 2. Now answer the following question.
To which period does this element belong?
Chapter: [0.02] Periodic Classification of Elements
Complete the following table:
| Chemical Reactions | Name reaction type |
| \[\ce{C(s) + O2(g) -> CO2(g)}\] | (a) ________ |
| \[\ce{Zn(s) + CuSO4(aq) ->}\] (b) _________ | Displacement reaction |
| \[\ce{2Cu(s) + O2(g) ->[heat] 2CuO(s)}\] | (c) _________ |
| \[\ce{C(s) + 2H2(g) -> CH4(g)}\] | (d) _________ |
| \[\ce{CaO(s) + H2O(l) -> Ca(OH)2(aq) + heat}\] | (e) _________ |
| \[\ce{CaCO3(s) + Heat ->}\] (f) _________ | Endothermic Reaction |
Chapter: [0.03] Chemical Reactions and Equations
Answer the questions based on the figure below:
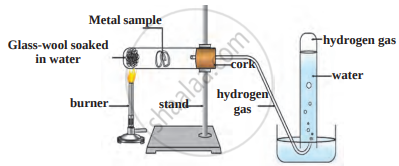
- Which reaction is shown in the figure?
- Which gas is evolved in the reaction?
- Give an example of reactants that rapidly show this reaction. Give equations.
- Give an example of reactants that do not react rapidly.
- In what condition will reactants of (c) part react? Give equation.
Chapter: [0.08] Metallurgy
Observe the following diagram and answer the questions given below:

- Identify the above diagram.
- Write the principle on which the above appliance works.
- Write the working of the above appliance.
- Write the use of the above appliance.
Chapter: [0.04] Effects of Electric Current
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 10th Standard Board Exam Science and Technology 1 with solutions 2024 - 2025
Previous year Question paper for Maharashtra State Board 10th Standard Board Exam -2025 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science and Technology 1, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 10th Standard Board Exam.
How Maharashtra State Board 10th Standard Board Exam Question Paper solutions Help Students ?
• Question paper solutions for Science and Technology 1 will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
