Advertisements
Advertisements
Represent diagrammatically how the incident planar wavefronts of wavelength λ pass through an aperture of size d, when d is approximately equal to λ.
Concept: undefined > undefined
During Rutherford’s gold foil experiment, it was observed that most of the α-particles did not deflect. However, some showed a deflection of 180°.
What hypothesis was made to justify the deflection of α-particle by 180°?
Concept: undefined > undefined
Advertisements
Represent diagrammatically how the incident planar wavefronts of wavelength λ pass through an aperture of size d, when d is approximately equal to λ.
Concept: undefined > undefined
In Bohr’s model of the hydrogen atom, the radius of the first orbit of an electron is r0 . Then, the radius of the third orbit is:
a) `r_0/9`
b) `r_0`
c) `3r_0`
d) `9r_0`
Concept: undefined > undefined
What is the ratio of the speed of gamma rays to that of radio waves in a vacuum?
Concept: undefined > undefined
Draw separate energy band diagram for conductors, semiconductors and insulators and
label each of them.
Concept: undefined > undefined
A fish which is at a depth of l2 em .in water `(mu = 4/3)` is viewed by an observer on the bank of a lake. Its apparent depth as observed: by the observer is:
a) 3 cm
b) 9 cm
c) 12 cm
d) 16 cm
Concept: undefined > undefined
if `E_p` and `E_k` represent potential energy and kinetic energy respectively, of an orbital electron, then, according to B9hr's theory:
a)`E_k = -E_p"/"2`
b) `E_k = -E_p`
c) `E_k = -2E_p`
d) `E_k = 2E_p`
Concept: undefined > undefined
How are electric vector `(vec "E")`, magnetic vector `(vec "B")` and velocity vector `(vec "C")` oriented in an electromagnetic wave?
Concept: undefined > undefined
Whatis the difference between analogue signal and. digital signal?
Concept: undefined > undefined
On the basis of Huygens Wave theory of light, show that angle of reflection is equal to the angle of incidence. You must draw a labelled diagram for this derivation
Concept: undefined > undefined
In an experiment of the photoelectric effect, the graph of maximum kinetic energy EK of the emitted photoelectrons versus the frequency v of the incident light is a straight line AB shown in Figure 6 below:
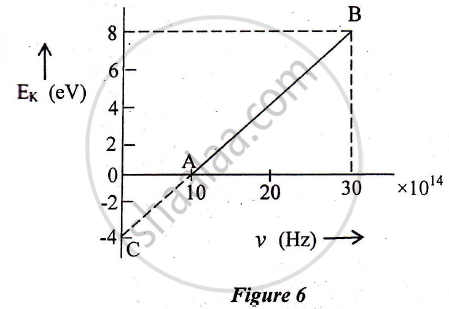
Find:
1) Threshold frequency of the metal
2) The work function of the metal.
3) Stopping potential for the photoelectrons emitted by the light of frequency `v = 30 xx 10^14 Hz`
Concept: undefined > undefined
On the basis of Bohr's theory, derive an expression for the radius of the nth orbit of an electron of the hydrogen atom.
Concept: undefined > undefined
The energy associated with light of which of the following colours is minimum :
Concept: undefined > undefined
This is an example of step-up transformer.
Concept: undefined > undefined
Define frequency modulation and state any one advantage of frequency modulation (FM) over amplitude modulation (AM).
Concept: undefined > undefined
State any two Bohr’s postulates and write the energy value of the ground state of the hydrogen atom.
Concept: undefined > undefined
In which of the following systems will the wavelength corresponding to n = 2 to n = 1 be minimum?
Concept: undefined > undefined
Calculate the momentum of a photon of energy 6 x I 0-19 J.
Concept: undefined > undefined
According to Bohr, 'Angular momentum of an orbiting electron is quantized'. What is meant by this statement?
Concept: undefined > undefined
