Advertisements
Advertisements
Why are pentahalides more covalent than trihalides?
Concept: undefined > undefined
Why is BiH3 the strongest reducing agent amongst all the hydrides of Group 15 elements?
Concept: undefined > undefined
Advertisements
Considering the parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy, compare the oxidising power of F2 and Cl2.
Concept: undefined > undefined
Discuss the general characteristics of Group 15 elements with reference to their electronic configuration, oxidation state, atomic size, ionisation enthalpy and electronegativity.
Concept: undefined > undefined
Discuss the trends in chemical reactivity of group 15 elements.
Concept: undefined > undefined
Why does R3P=O exist but R3N=O does not (R = alkyl group)?
Concept: undefined > undefined
Nitrogen exists as diatomic molecule and phosphorus as P4. Why?
Concept: undefined > undefined
Write main differences between the properties of white phosphorus and red phosphorus.
Concept: undefined > undefined
Why does nitrogen show catenation properties less than phosphorus?
Concept: undefined > undefined
Arrange the following in the increasing order of property mentioned :
NH3, PH3, AsH3, SbH3, BiH3 (Base strength)
Concept: undefined > undefined
Classify the following as primary, secondary and tertiary alcohol:
H2C = CH – CH2OH
Concept: undefined > undefined
Classify the following as primary, secondary and tertiary alcohol:
CH3 – CH2 – CH2 – OH
Concept: undefined > undefined
Classify the following as primary, secondary and tertiary alcohol:
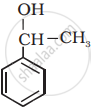
Concept: undefined > undefined
Classify the following as primary, secondary and tertiary alcohol:
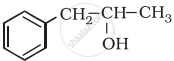
Concept: undefined > undefined
Classify the following as primary, secondary and tertiary alcohol:
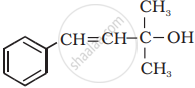
Concept: undefined > undefined
Write the structure of 3-methyl butanal
Concept: undefined > undefined
What happens if external potential applied becomes greater than E°cell of electrochemical cell?
Concept: undefined > undefined
Write two differences between an ideal solution and a non-ideal solution.
Concept: undefined > undefined
Account for the following : Nitrogen does not form pentahalide.
Concept: undefined > undefined
Write the structure of 2-methylbutanal.
Concept: undefined > undefined
