Advertisements
Advertisements
प्रश्न
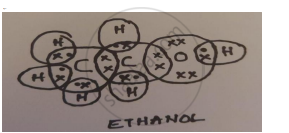
A carbon compound ‘A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
- Identify ‘A’ and draw its electron dot structure.
- Give the molecular formulae of any two homologues of ‘A’.
उत्तर
- Ethanol; C2H5OH
- CH3OH and C3H7OH are homologues of ethanol
OR
CH4O and C₃H₈O
APPEARS IN
संबंधित प्रश्न
Fill in the following blank with suitable word:
Carbon compounds have usually ... melting points and boiling points because they are ...... in nature.
The molecular formula of a homologue of butane is:
(a) C4H8
(b) C3H6
(c) C4H6
(d) C3H8
An organic compound having the molecular formula C3H6O can exist in the form of two isomers A and B having different functional groups. The isomer A is a liquid which is used as a solvent for nail polish. The isomer B is also a liquid. An aqueous solution of one of the lower homologues of B is used for preserving biological specimens in the laboratory
(a) What is compound A?
(b) Write the electron-dot structure of A.
(c) What is compound B?
(d) Write the electron-dot structure of B.
(e) Name the lower homologue of compound B which is used in preserving biological specimens.
Give the abbreviated formula of the third member of the alcohol.
 .
.
Give the names of the first four members of the homologous series of
alkynes.
The general formula of Alkane is _________________
Saturated hydrocarbon : Single bond : : Unsaturated hydrocarbon : _______
Find the odd one out and give its explanation.
Which of the following pairs can be the successive members of a homologous series?
