Advertisements
Advertisements
प्रश्न
Assertion: Λm for weak electrolytes shows a sharp increase when the electrolytic solution is diluted.
Reason: For weak electrolytes degree of dissociation increases with dilution of solution.
विकल्प
Both assertion and reason are true and the reason is the correct explanation of assertion.
Both assertion and reason are true and the reason is not the correct explanation of assertion.
Assertion is true but the reason is false.
Both assertion and reason are false.
Assertion is false but reason is true.
उत्तर
Both assertion and reason are true and the reason is the correct explanation of assertion.
Explanation:
Weak electrolytes dissociate partially in concentrated solution. On dilution, their degree of dissociation increases hence their Am increases sharply.
APPEARS IN
संबंधित प्रश्न
State Kohlrausch Law
10.0 grams of caustic soda when dissolved in 250 cm3 of water, the resultant gram molarity of solution is _______.
(A) 0.25 M
(B) 0.5 M
(C) 1.0 M
(D) 0.1 M
Calculate the degree of dissociation (α) of acetic acid if its molar conductivity (Λm) is 39.05 S cm2 mol−1.
Given λ°(H+) = 349.6 S cm2 mol−1 and λ°(CH3COO−) = 40.9 S cm2 mol−1
A steady current of 2 amperes was passed through two electrolytic cells X and Y connected in series containing electrolytes FeSO4and ZnSO4 until 2.8g of Fe deposited at the cathode of cell X. How long did the current flow? Calculate the mass of Zn deposited at the cathode of cell Y.
(Molar mass: Fe=56g mol-1,Zn=65.3g mol-1,1F=96500C mol-1)
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2) following curves are obtained for two electrolytes A and B : 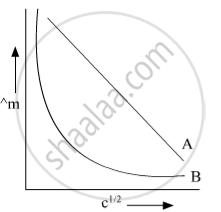
Answer the following:
(i) predict the nature of electrolytes A and B.
(ii) What happens on the extrapolation of ∧m to concentration approaching for electrolytes A and B?
Why on dilution the m Λm of \[\ce{CH3COOH}\] increases very fast, while that of \[\ce{CH3COONa}\] increases gradually?
Assertion: Copper sulphate can be stored in zinc vessel.
Reason: Zinc is less reactive than copper.
Consider figure and answer the question to given below.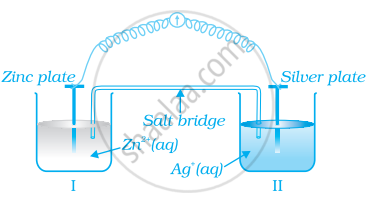
How will the concentration of Zn2+ ions and Ag+ ions be affected after the cell becomes ‘dead’?
The variation of molar conductivity with concentration of an electrolyte (X) m aqueous solution is shown in the given figure.
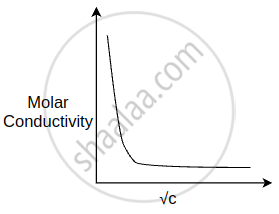
The electrolyte X is ______.
Which of the following solutions will have the highest conductivity at 298 K?
