Advertisements
Advertisements
प्रश्न
Why on dilution the m Λm of \[\ce{CH3COOH}\] increases very fast, while that of \[\ce{CH3COONa}\] increases gradually?
उत्तर
In the case of \[\ce{CH3COOH}\], which is a weak electrolyte, the number of ions increase on dilution due to an increase in degree of dissociation.
\[\ce{CH3COOH + H2O ⇌ CH3COO^{-} (aq) + H3O^{+}}\]
In the case of strong electrolyte such as \[\ce{CH3COONa}\], the number of ions remains the same but the interionic attraction decreases.
संबंधित प्रश्न
The molar conductivity of 0.025 mol L−1 methanoic acid is 46.1 S cm2 mol−1. Calculate its degree of dissociation and dissociation constant. Given \[\ce{λ^0_{(H^+)}}\] = 349.6 S cm2 mol−1 and \[\ce{λ^0_{(HCOO^-)}}\] = 54.6 S cm2 mol−1.
Conductivity of 0.00241 M acetic acid is 7.896 × 10−5 S cm−1. Calculate its molar conductivity and if `∧_"m"^0` for acetic acid is 390.5 S cm2 mol−1, what is its dissociation constant?
The conductivity of 0.02M AgNO3 at 25°C is 2.428 x 10-3 Ω-1 cm-1. What is its molar
conductivity?
Write mathematical expression of molar conductivity of the given solution at infinite dilution.
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2), following curves are obtained for two electrolytes A and B:
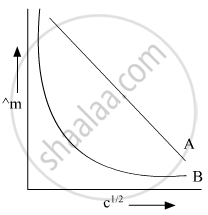
Answer the following:
(i) Predict the nature of electrolytes A and B.
(ii) What happens on extrapolation of ∧m to concentration approaching zero for electrolytes A and B?
Which of the statements about solutions of electrolytes is not correct?
\[\ce{Λ^0_m}_{(NH_4OH)}\] is equal to ______.
Assertion: Copper sulphate can be stored in zinc vessel.
Reason: Zinc is less reactive than copper.
Given below are two statements:
Statements I: The limiting molar conductivity of KCl (strong electrolyte) is higher compared to that of CH3COOH (weak electrolyte).
Statement II: Molar conductivity decreases with decrease in concentration of electrolyte.
In the light of the above statements, choose the most appropriate answer from the options given below:
The unit of molar conductivity is ______.
