Advertisements
Advertisements
प्रश्न
Conductivity of 2 × 10−3 M methanoic acid is 8 × 10−5 S cm−1. Calculate its molar conductivity and degree of dissociation if `∧_"m"^0` for methanoic acid, is 404 S cm2 mol−3.
उत्तर
Given: Conductivity (K) = 8 × 10−5 S cm−1
Molarity (M) = 2 × 10−3 M
Formula:
Molar conductivity, `∧_"m"^"c" = ("K" xx 1000)/"C"`
Degree of dissociation, α = `∧_"m"^"c"/∧_"m"^0`
`∧_"m"^0 = ("K" xx 1000)/"C"` S cm2 mol−1
= `(8 xx 10^-5 xx 1000)/(2 xx 10^-3)`
= 40 S cm2 mol−1
`∝` = `(∧_"m"^"c")/(∧_"m"^0)`
= `40/404`
= 0.1
APPEARS IN
संबंधित प्रश्न
Define "Molar conductivity".
Resistance of conductivity cell filled with 0.1 M KCl solution is 100 ohms. If the resistance of the same cell when filled with 0.02 M KCl solution is 520 ohms, calculate the conductivity and molar conductivity of 0.02 M KCl solution. [Given: Conductivity of 0.1 M KCl solution is 1.29 S m-1 .]
The conductivity of 0.02M AgNO3 at 25°C is 2.428 x 10-3 Ω-1 cm-1. What is its molar
conductivity?
Conductivity always decreases with decrease in concentration both, for weak and strong electrolytes because of the fact that ____________.
Solutions of two electrolytes ‘A’ and ‘B’ are diluted. The Λm of ‘B’ increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Justify your answer.
When acidulated water (dil.H2SO4 solution) is electrolysed, will the pH of the solution be affected? Justify your answer.
Write the cell reaction of a lead storage battery when it is discharged. How does the density of the electrolyte change when the battery is discharged?
Consider figure and answer the question to given below.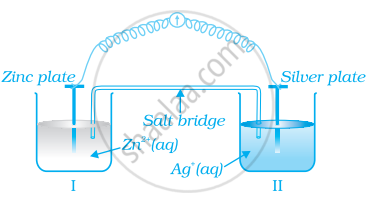
How will the concentration of Zn2+ ions and Ag+ ions be affected after the cell becomes ‘dead’?
The molar conductance of NaCl, HCl, and CH3COONa at infinite dilution are 126.45, 426.16, and 91.0 S cm2 mol−1 respectively. The molar conductance of CH3COOH at infinite dilution is. Choose the right option for your answer.
Assertion (A) : Conductivity decreases with decrease in concentration of electrolyte.
Reason (R) : Number of ions per unit volume that carry the current in a solution decreases on dilution.
