Advertisements
Advertisements
प्रश्न
How will you bring about the following conversions?
isopropyl alcohol to acetone
उत्तर
When isopropyl alcohol is heated at 573 K in presence of Cu acetone is formed.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[Cu/573K][Oxidation] CH3 - C - CH3}\\
\phantom{...}|\phantom{..........................}||\phantom{...}\\
\phantom{}\ce{\underset{\text{Isopropyl alcohol}}{OH}\phantom{...............}\underset{\text{Acetone}}{O}}\end{array}\]
संबंधित प्रश्न
Answer in one sentence/ word.
Hydroboration-oxidation of propene gives _________.
Answer in one sentence/ word.
Write the IUPAC name of alcohol having molecular formula C4H10O which is resistant towards oxidation.
Answer in brief.
Explain why phenol is more acidic than ethyl alcohol.
Write two points of difference between the properties of phenol and ethyl alcohol.
Give the reagents and conditions necessary to prepare phenol from Chlorobenzene.
Answer in brief.
Give the equations of the reactions for the preparation of phenol from isopropyl benezene.
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
What is the action of following on phenol at low temperature?
- dil. HNO3
- conc. H2SO4
- Br2/CS2
Write chemical reactions to convert phenol into salicyaldehyde. Write the name of the reaction. What happens when if CCl4 is used instead of CHCl3 in the above reaction.
Which of following elements does not react with hot concentrated sulphuric acid?
The number of isomeric alcohols possible with the formula C4H10O is ____________.
Identify 'A' and 'B' in the following series of reactions.
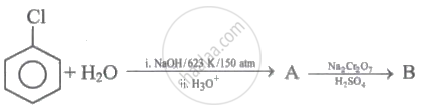
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
Phenoxide ion is more stable than phenol due to the ____________.
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.
The most resistant alcohol towards oxidation reaction is:
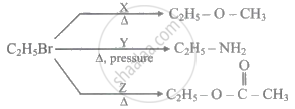
Identify reagents X, Y and Z.
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
Which of the following conversion explains the acidic nature of alcohols?
Which following reagent is used to obtain alkene from alcohol?
Which among the following is allylic secondary alcohol?
The compound which reacts fastest with Lucas reagent at room temperature is ______.
Identify the compound amongst the following of which 0.1 M aqueous solution has highest boiling point.
Which of the following compounds has lowest boiling point?
What is the product formed when aniline is treated with \[\ce{NaNO2 + HCl}\] previous to hydrolysis?
Which of the following compounds reacts immediately with Lucas reagent?
The major product obtained in the following reaction is

Write the chemical reaction when hot copper is treated with Vapours of 1° (primary) alcohol.
