Advertisements
Advertisements
प्रश्न
If an average person jogs, hse produces 14.5 × 103 cal/min. This is removed by the evaporation of sweat. The amount of sweat evaporated per minute (assuming 1 kg requires 580 × 103 cal for evaparation) is ______.
विकल्प
0.25 kg
2.25 kg
0.05 kg
0.20 kg
उत्तर
If an average person jogs, hse produces 14.5 × 103 cal/min. This is removed by the evaporation of sweat. The amount of sweat evaporated per minute (assuming 1 kg requires 580 × 103 cal for evaparation) is 0.25 kg.
Explanation:
The rate of bum calories is equivalent to sweat produced. Then, the amount of sweat evaporated/minute
= `"Sweat produced/minute"/"Number of calories required for evaporation/kg"`
= `"Calories produced (heat produced) per minute"/"Latent heat (in cal/kg)"`
= `(14.5 xx 10^3)/(580 xx 10^3)`
= `145/580`
= 0.25 kg
APPEARS IN
संबंधित प्रश्न
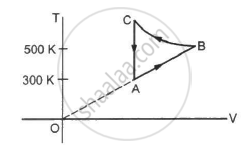
Consider the cyclic process ABCA, shown in figure, performed on a sample of 2.0 mol of an ideal gas. A total of 1200 J of heat is withdrawn from the sample in the process. Find the work done by the gas during the part BC.
Choose the correct option.
Which of the following is an example of the first law of thermodynamics?
Two moles of an ideal gas is expanded isothermally and reversibly at 300 K from 1 L to 10 L. The enthalpy change in kJ is ______.
Based on first law of thermodynamics which of the following is correct.
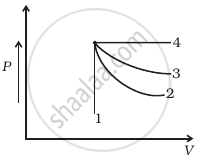
An ideal gas undergoes four different processes from the same initial state (figure). Four processes are adiabatic, isothermal, isobaric and isochoric. Out of 1, 2, 3 and 4 which one is adiabatic.
Consider that an ideal gas (n moles) is expanding in a process given by P = f(V), which passes through a point (V0, P0). Show that the gas is absorbing heat at (P0, V0) if the slope of the curve P = f(V) is larger than the slope of the adiabat passing through (P0, V0).
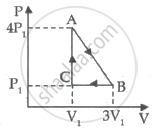
An ideal gas is taken through series of changes ABCA. The amount of work involved in the cycle is ______.
The first law of thermodynamics for isothermal process is ______.
The amount of work done in increasing the voltage across the plates of capacitor from 5 V to 10 V is W. The work done in increasing it from 10 V to 15 V will be ______.
Consider the cyclic process ABCA on a sample of 2.0 mol of an ideal gas as shown in following figure. The temperature of the gas at A and B are 300 K and 500 K respectively. A total of 1200 J heat is withdrawn from the sample in this process. Find the work done by the gas in part BC. (R = 8.3 J/mol K)
