Advertisements
Advertisements
Question
If an average person jogs, hse produces 14.5 × 103 cal/min. This is removed by the evaporation of sweat. The amount of sweat evaporated per minute (assuming 1 kg requires 580 × 103 cal for evaparation) is ______.
Options
0.25 kg
2.25 kg
0.05 kg
0.20 kg
Solution
If an average person jogs, hse produces 14.5 × 103 cal/min. This is removed by the evaporation of sweat. The amount of sweat evaporated per minute (assuming 1 kg requires 580 × 103 cal for evaparation) is 0.25 kg.
Explanation:
The rate of bum calories is equivalent to sweat produced. Then, the amount of sweat evaporated/minute
= `"Sweat produced/minute"/"Number of calories required for evaporation/kg"`
= `"Calories produced (heat produced) per minute"/"Latent heat (in cal/kg)"`
= `(14.5 xx 10^3)/(580 xx 10^3)`
= `145/580`
= 0.25 kg
APPEARS IN
RELATED QUESTIONS
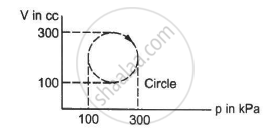
Calculate the heat absorbed by a system in going through the cyclic process shown in figure.
ΔU is equal to ____________ work.
Consider a cycle followed by an engine (Figure)
1 to 2 is isothermal
2 to 3 is adiabatic
3 to 1 is adiabatic
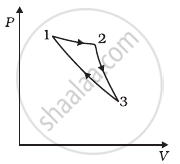
Such a process does not exist because ______.
- heat is completely converted to mechanical energy in such a process, which is not possible.
- mechanical energy is completely converted to heat in this process, which is not possible.
- curves representing two adiabatic processes don’t intersect.
- curves representing an adiabatic process and an isothermal process don’t intersect.
Is it possible to increase the temperature of a gas without adding heat to it? Explain.
A cycle followed by an engine (made of one mole of an ideal gas in a cylinder with a piston) is shown in figure. Find heat exchanged by the engine, with the surroundings for each section of the cycle. (Cv = (3/2)R)
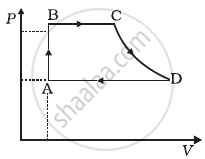
- AB : constant volume
- BC : constant pressure
- CD : adiabatic
- DA : constant pressure
Write the mathematical equation for the first law of thermodynamics for:
Isothermal process
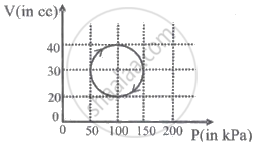
A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is ______.
The first law of thermodynamics for isothermal process is ______.
One mole of an ideal gas is allowed to expand reversibly and adiabatically from a temperature of 27°C. If the work done during the process is 3 kJ, the final temperature will be equal to ______.
(Cv = 20 JK−1)
Calculate work done when 2 moles of ideal gas expands by 5 dm3 isothermally at pressure 1.2 bar.
