Advertisements
Advertisements
Question
An ideal gas undergoes four different processes from the same initial state (figure). Four processes are adiabatic, isothermal, isobaric and isochoric. Out of 1, 2, 3 and 4 which one is adiabatic.
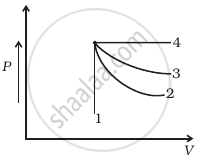
Options
4
3
2
1
Solution
2
Explanation:
For curve 4 pressure is constant, so this is an isobaric process.

For curve 1, volume is constant, so it is an isochoric process. Between curves 3 and 2, curve 2 is steeper, so it is adiabatic and 3 is isothermal.
APPEARS IN
RELATED QUESTIONS
Calculate the increase in the internal energy of 10 g of water when it is heated from 0°C to 100°C and converted into steam at 100 kPa. The density of steam = 0.6 kg m−3. Specific heat capacity of water = 4200 J kg−1 °C−1 and the latent heat of vaporization of water = 2.25 × 10 6J kg−1.
Choose the correct option.
Which of the following is an example of the first law of thermodynamics?
10 kg of four different gases (Cl2, CH4, O2, N2) expand isothermally and reversibly from 20 atm to 10 atm. The order of amount of work will be ____________.
"The mass and energy both are conserved in an isolated system", is the statement of ______.
The process, in which no heat enters or leaves the system, is termed as ____________.
104 J of work is done on a certain volume of a gas. If the gas releases 125 kJ of heat, calculate the change in internal energy of the gas.
One mole of an ideal gas is initially kept in a cylinder with a movable frictionless and massless piston at pressure of 1.01MPa, and temperature 27°C. It is then expanded till its volume is doubled. How much work is done if the expansion is isobaric?
A monoatomic gas at 27°C is adiabatically compressed to 80% of its initial volume. Find the final temperature of the gas.
Obtain an expression for the workdone by a gas in an isothermal process.
Define isochoric process
