Advertisements
Advertisements
प्रश्न
In the series Sc (Z = 21) to Zn (Z = 30), the enthalpy of atomization of zinc is the lowest, i.e., 126 kJ mol−1. Why?
उत्तर
The electrons of the 3d orbitals of zinc are not used in bonding, whereas the electrons of the d orbitals of all the remaining metals of the 3d series are used in forming bonds. Hence the value of the atomization enthalpy of zinc is the lowest in the series.
APPEARS IN
संबंधित प्रश्न
What are interstitial compounds?
Account for the following:
Mn shows the highest oxidation state of +7 with oxygen but with fluorine, it shows oxidation state of +4.
Give reasons: Transition metals show variable oxidation states.
What are the transition elements? Write two characteristics of the transition elements.
Calculate the ‘spin only’ magnetic moment of \[\ce{M^{2+}_{ (aq)}}\] ion (Z = 27).
Highest oxidation state of manganese in fluoride is \[\ce{+4 (MnF4)}\] but highest oxidation state in oxides is \[\ce{+7 (Mn2O7)}\] because ______.
Match the solutions given in Column I and the colours given in Column II.
| Column I (Aqueous solution of salt) |
Column II (Colour) |
| (i) \[\ce{FeSO2.7H2O}\] | (a) Green |
| (ii) \[\ce{NiCl2.4H2O}\] | (b) Light pink |
| (iii) \[\ce{MnCl2.4H2O}\] | (c) Blue |
| (iv) \[\ce{CoC12,6H2O}\] | (d) Pale green |
| (v) \[\ce{Cu2 Cl2}\] | (e) Pink |
| (f) Colourless |
Identify A to E and also explain the reactions involved.
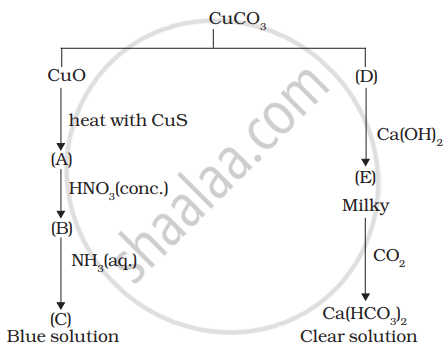
A violet compound of manganese (A) decomposes on heating to liberate oxygen and compounds (B) and (C) of manganese are formed. Compound (C) reacts with KOH in the presence of potassium nitrate to give compound (B). On heating compound (C) with conc. \[\ce{H2SO4}\] and \[\ce{NaCl}\], chlorine gas is liberated and a compound (D) of manganese along with other products is formed. Identify compounds A to D and also explain the reactions involved.
The product of oxidation of I– with \[\ce{MnO^{-}4}\] in alkaline medium is:-
Which of the following ions will exhibit colour in aqueous solution?
A complex in which dsp2 hybridisation takes place is
Why is the `"E"_(("V"^(3+)//"V"^(2+)))^"o"` value for vanadium comparatively low?
In the ground state of atomic Fe (Z = 26), the spin-only magnetic moment is ______ × 10-1 BM.
(Round off to the nearest integer).
[Given: `sqrt3 = 1.73, sqrt2 = 1.41`]
Which of the following characteristics of transition metals is associated with their catalytic activity?
The given graph shows the trends in melting points of transition metals:

Explain the reason why Cr has the highest melting point and manganese (Mn) has a lower melting point.
A transition element X has an electronic configuration [Ar]4s23d3. Predict its likely oxidation states.
Give a reason for the following:
Zinc, cadmium and mercury are considered as d-block elements but not regarded as transition elements.
Give a reason for the following:
Transition metals possess a great tendency to form complex compounds.
