Advertisements
Advertisements
प्रश्न
Methane molecule is non-polar molecule. Explain.
उत्तर
During the formation of a non-polar covalent bond between two similar atoms or dissimilar atoms, the atoms involved in sharing share the electrons equally. The molecule of methane has four carbon-hydrogen single covalent bonds. It is a non-polar covalent compound as the electrons are shared by the carbon and hydrogen atoms equally and hence the shared pair lies between the atoms at an equal distance from both carbon and hydrogen atom.
\[\begin{array}{cc}
\ce{H}\\
|\\
\ce{H - C - H}\\
|\\
\ce{H}\\
\end{array}\]
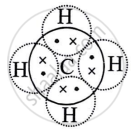 Methane
Methane
APPEARS IN
संबंधित प्रश्न
Give the formula of the compound that would be formed by the combination of the following pair of elements:
K and H
The atomic numbers of four elements P, Q, R and S are 6, 10, 12 and 17 respectively. Which two elements can combine to form a covalent compound?
(a) P and R
(b) Q and S
(c) P and S
(d) R and S
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
What type of bond is formed between Y and Z.
Which of the following is a common characteristic of a covalent compound?
Draw an electron dot diagram to show the formation of the following compound.
Methane
Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
The type of bonding in X will be ______.
From the following hydrocarbon _______ is the cyclic hydrocarbon.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\ |\phantom{....}|\phantom{....}|\\\ce{H - C - C - C - H}\\ |\phantom{....}|\phantom{....}|\\\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H} \end{array}\] |
Which of the following is the formula of Butanoic acid?
