Advertisements
Advertisements
प्रश्न
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume :
SO2Cl2 (g) → SO2 (g) + Cl2 (g)
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.4 |
| 2 | 100 | 0.7 |
Calculate the rate constant.
(Given : log 4 = 0.6021, log 2 = 0.3010)
उत्तर १
The given reaction is as follows:
SO2Cl2 (g) → SO2(g)+Cl2(g)
Also given:
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.4 |
| 2 | 100 | 0.7 |
The rate constant k can be calculated as follows:
`k=2.303/tlog `
when t= 100s, k = `2.303/(100s)log`
`k = 2.303/(100s)log`
`k=2.303/(100s) log 4`
`k = 2.303/(100s)xx0.6021`
k = 1.39 x 10-2s-1
Therefore, the rate constant is 1.39 x 10-2s-1
उत्तर २
The thermal decomposition of SO2Cl2 at a constant volume is represented by the following equation:
SO2Cl2→SO2(g) + Cl2(g)
At t =0 P0 0 0
At t=t P0 −p p p
After time t, total pressure is given as:
Pt =(P0−p) + p + p
Pt = P0 + p
This, on rearrangement, gives:
p = Pt − P0
∴ P0−p=P0−(Pt−P0)= 2P0 − Pt
For the first-order reaction, we have:
`k=2.303/tlog P_0/(P_0 - p)`
`=2.303/tlog P_0/(P_0 - p)`
t = 100 s
`k = 2.303/100log 0.42/(0.4 - 0.7) = 1.386 xx 10^(-2)s-1`
APPEARS IN
संबंधित प्रश्न
For a reaction : 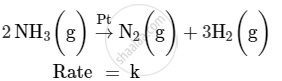
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
Write resonating structures of ozone.
Write the principle behind the following methods of refining:
Hydraulic washing
Define the following terms:
Pseudo first-order reaction
Why is the probability of reaction with molecularity higher than three very rare?
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
Assertion: Order and molecularity are same.
Reason: Order is determined experimentally and molecularity is the sum of the stoichiometric coefficient of rate determining elementary step.
For a reaction A + B → products, the rate law is given by: r = `K[A]^(1/2)`. What is the order of reaction?
For a reaction \[\ce{Cl2l(g) + 2No(g) -> 2NaCl(g)}\] the rate law is expressed as rate= K[Cl2] [No]2 what is the order of the reaction?
Which of the following statement is true?
