Advertisements
Advertisements
प्रश्न
The passage of an electric current through a solution causes ______ effects.
उत्तर
The passage of an electric current through a solution causes chemical effects.
Explanation:
The passage of an electric current through a conducting solution causes chemical reactions. As a result, bubbles of gas may be formed on the electrodes. Deposits of metal may be seen on electrodes. Changes in the colour of solutions may occur. The reaction would depend on what solution and electrodes are used. These are some of the chemical effects of the electric current.
APPEARS IN
संबंधित प्रश्न
If you pass current through copper sulphate solution, copper gets deposited on the plate connected to the ______ terminal of the battery.
The process that you saw in the image is used for the purification of copper. A thin plate of pure copper and a thick rod of impure copper are used as electrodes. Copper from impure rod is sought to be transferred to the thin copper plate. Which electrode should be attached to the positive terminal of the battery and why?
Which of the following metals is used in electroplating to make objects appear shining?
One of the most common applications of chemical effect of electric current is ______.
Paheli set up an experiment using liquid A in the beaker as shown in the figure. She observed that the bulb glows. Then, she replaced the liquid A by another liquid B. This time the bulb did not glow. Boojho suggested replacing the bulb by an LED. They observed that the LED glows. Explain.
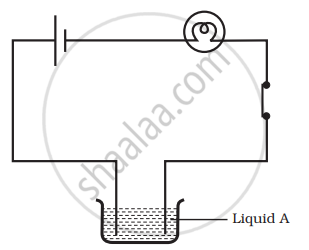
Why is tin electroplated on iron to make cans used for storing food?
In the circuit given in the figure,
Boojho observed that copper is deposited on the electrode connected to the negative terminal of the battery.
Paheli tried to repeat the same experiment. But she could find only one copper plate. Therefore, she took a carbon rod as negative electrode. Will copper be still deposited on the carbon rod? Explain your answer.
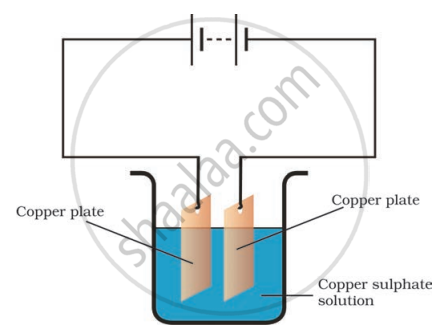
A chemical reaction happens when electricity passes through various conducting liquids.
The most common industrial application of chemical effects of electric current is ______.
______ is used in the extraction and purification of metals.
