Advertisements
Advertisements
Question
The passage of an electric current through a solution causes ______ effects.
Solution
The passage of an electric current through a solution causes chemical effects.
Explanation:
The passage of an electric current through a conducting solution causes chemical reactions. As a result, bubbles of gas may be formed on the electrodes. Deposits of metal may be seen on electrodes. Changes in the colour of solutions may occur. The reaction would depend on what solution and electrodes are used. These are some of the chemical effects of the electric current.
APPEARS IN
RELATED QUESTIONS
The process of depositing a layer of any desired metal on another material by means of electricity is called ______.
In an electrolyte the current is due to the flow of ______.
Boojho and Paheli performed experiments taking similar bulbs and cells but two different solutions A and B as shown in the figure.
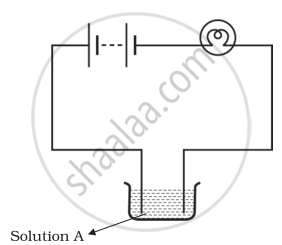 |
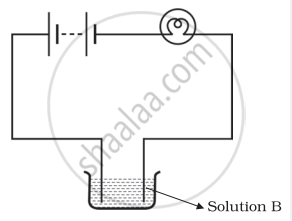 |
| Boojho's Experiment | Paheli's Experiment |
| (A) | (B) |
They found that the bulb in the set up A glows more brightly as compared to that of the set up B. You would conclude that
Boojho’s uncle has set up an electroplating factory near his village. He should dispose off, the waste of the factory
When electric current is passed through a conducting solution, there is a change of colour of the solution. This indicates
The object to be electroplated is taken as ______ electrode.
One of the most common applications of chemical effect of electric current is ______.
Electroplating of ______ is done on objects like water taps and cycle bell to give them a shiny appearance.
Paheli wants to deposit silver on an iron spoon. She took silver nitrate (AgNO3) solution in a beaker and set up a simple circuit for electroplating. Which terminal of the battery should the spoon be connected to? What material should the other electrode be made of?
The most common industrial application of chemical effects of electric current is ______.
