Advertisements
Advertisements
प्रश्न
The passage of an electric current through a solution causes ______ effects.
उत्तर
The passage of an electric current through a solution causes chemical effects.
Explanation:
The passage of an electric current through a conducting solution causes chemical reactions. As a result, bubbles of gas may be formed on the electrodes. Deposits of metal may be seen on electrodes. Changes in the colour of solutions may occur. The reaction would depend on what solution and electrodes are used. These are some of the chemical effects of the electric current.
APPEARS IN
संबंधित प्रश्न
Name three liquids, which when tested in the manner shown in Figure, may cause the magnetic needle to deflect.
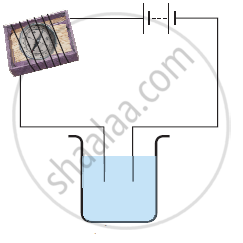
In an electrolyte the current is due to the flow of ______.
When electric current is passed through a conducting solution, there is a change of colour of the solution. This indicates
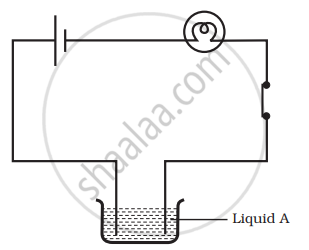
Paheli set up an experiment using liquid A in the beaker as shown in the figure. She observed that the bulb glows. Then, she replaced the liquid A by another liquid B. This time the bulb did not glow. Boojho suggested replacing the bulb by an LED. They observed that the LED glows. Explain.
Paheli wants to deposit silver on an iron spoon. She took silver nitrate (AgNO3) solution in a beaker and set up a simple circuit for electroplating. Which terminal of the battery should the spoon be connected to? What material should the other electrode be made of?
You are provided with a magnetic compass, an empty matchbox, a battery of two cells and connecting wires. Using these objects, how will you make a tester for testing an electric circuit? Draw the necessary circuit diagram and explain.
The terminal which is connected to a positive terminal of a battery is called ______.
Liquids that conduct electricity are the solutions of ______.
State some advantages of electroplating.
What are anodes and cathodes?
