Advertisements
Advertisements
प्रश्न
The usefulness of a fertilizer depends upon percentage of nitrogen present in it. Find which of the following is a better fertilizer:
(a) Ammonium nitrate [NH4NO3]
(b) Ammonium phosphate [(NH4)3PO4 (N=14,H=1,O=16,P=31)
उत्तर
(a) Percentage of nitrogen in Ammonium nitrate [NH4NO3]:
(N)2 + (H)4 + (O)3
14 x 2 + 1 x 4 + 3 x 16 = 80.
So, the molecular mass of NH4NO3 = 80.
80 by weight of NH4NO3 contain 28 parts by weight of nitrogen .
100 parts will contain = 28 x 100 /80 = 35‰
So, the percentage composition of nitrogen in Ammonium nitrate is 3535‰
(b) Percentage of nitrogen in Ammonium phosphate [(NH4)3PO4]:
(N)3 + (H)12 +P + (O)4
14 x 3 + 1 x 12+ 31 + 16 x 4 = 149 .
So, the molecular mass of NH4NO3 = 149 .
149 by weight of (NH4)3PO4.contain 42 parts by weight of nitrogen .
100 parts will contain = 42 x 100 /149 = 28.18‰
So, the percentage of nitrogen in ammonium phosphate is 28.18‰
Since the percentage of nitrogen is more in Ammonium nitrate so it is a better fertilizer.
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
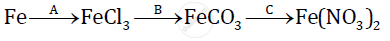
10g of NaCl solution is mixed with 17g of silver nitrate solution. Calculate the weight of silver chloride precipitated.
AgNO3 + NaCl → AgCI + NaNO3
What is the mass of nitrogen in 1000Kg of urea [CO(NH2)2] ?
[H = 1, C= 12, N= 14, O = 16]
Calculate the percentage of boron (B) in borax (Na2B4O7.10H2O). [H = 1, B = 11, O = 16, Na = 23],
answer correct to 1 decimal place.
If the relative molecular mass of A is 90, what is the molecular formula of A?
Calculate the relative molecular mass of:
CHCl3
Calculate the mass of nitrogen supplied to soil by 5 kg of urea [CO(NH2)2].
[O = 16; N = 14; C = 12; H = 1]
What do you understand by the statement that 'vapour density of carbon dioxide is 22'?
The atomic mass of Chlorine is 35.5. What is its vapour density?
The following question refers to one mole of chlorine gas.
If the relative atomic mass of chlorine is 35.5, what will be the mass of 1 mole of chlorine gas?
