Advertisements
Advertisements
प्रश्न
Three copper blocks of masses M1, M2 and M3 kg respectively are brought into thermal contact till they reach equilibrium. Before contact, they were at T1, T2, T3 (T1 > T2 > T3). Assuming there is no heat loss to the surroundings, the equilibrium temprature T is (s is specific heat of copper)
विकल्प
`T = (T_1 + T_2 + T_3)/3`
`T = (M_1T_1 + M_2T_2 + M_3T_3)/(M_1 + M_2 + M_3)`
`T = (M_1T_1 + M_2T_2 + M_3T_3)/(3(M_1 + M_2 + M_3))`
`T = (M_1T_1s + M_2T_2s + M_3T_3s)/(M_1 + M_2 + M_3)`
उत्तर
`T = (M_1T_1 + M_2T_2 + M_3T_3)/(M_1 + M_2 + M_3)`
Explanation:
Let the equilibrium temperature to be found be T. Now, we consider T to be greater than T1 and T2 but smaller than T3.
Since there is no loss of heat energy. Hence, we get,
Heat lost by M3 = Heat regained by M1 + Heat regained by M2 So, we get,
⇒ M3s (T3 – T) = M1s (T – T1) + M2s (T – T2)
Dividing both sides by s, we get,
⇒ M3 (T3 – T) = M1 (T – T1) + M2 (T – T2)
Opening the brackets and solving for T, we get,
⇒ M3T3 – M3T = M1T – M1T1 + M2T – M2T2
⇒ M3T3 + M1T1 + M2T2 = M1T + M2T + M3T
⇒ `T = (M_1T_1 + M_2T_2 + M_3T_3)/(M_1 + M_2 + M_3)`
APPEARS IN
संबंधित प्रश्न
A thermally insulated, closed copper vessel contains water at 15°C. When the vessel is shaken vigorously for 15 minutes, the temperature rises to 17°C. The mass of the vessel is 100 g and that of the water is 200 g. The specific heat capacities of copper and water are 420 J kg−1 K−1 and 4200 J kg−1 K−1 respectively. Neglect any thermal expansion. (a) How much heat is transferred to the liquid-vessel system? (b) How much work has been done on this system? (c) How much is the increase in internal energy of the system?
For an Isochoric process
The isothermal bulk modulus of a perfect gas at pressure P is numerically equal to ____________.
An ideal gas undergoes four different processes from the same initial state (figure). Four processes are adiabatic, isothermal, isobaric and isochoric. Out of 1, 2, 3 and 4 which one is adiabatic.
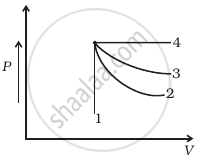
The first law of thermodynamics is concerned with the conservation of ______.
Mathematical equation of first law of thermodynamics for isochoric process is ______.
The first law of thermodynamics for isothermal process is ______.
An ideal gas having pressure p, volume V and temperature T undergoes a thermodynamic process in which dW = 0 and dQ < 0. Then, for the gas ______.
Define the isothermal process.
Define isochoric process
