Advertisements
Advertisements
प्रश्न
What happens when a cold and concentrated solution of sodium chloride reacts with ammonia and carbon dioxide? Write the chemical equation of the reaction which takes place.
उत्तर
Sodium hydrogencarbonate (baking soda) is produced when a cold and concentrated solution of sodium chloride (brine) reacts with ammonia and carbon dioxide.
NaCl + NH3 + H2O + CO2 → NaHCO3 + NH4Cl
APPEARS IN
संबंधित प्रश्न
What will happen if a solution of sodium hydrocarbonate is heated? Give the equation of the reaction involved.
Give two important uses of baking soda.
Which compound is used as an antacid in medicine : NaHCO3 or Na2CO3?
Complete and balance the following chemical equations:
`NaHCO_3` 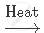
What is the common name of sodium hydrogencarbonate?
How does baking powder differ from baking soda?
What are the materials used for the preparation of bleaching powder?
(i) Name the products formed when sodium hydrogen carbonate is heated.
(ii) Write the chemical equation for the reaction involved in the above.
The chemical name of baking soda is ______.
Write the chemical formula.
Baking soda
