Advertisements
Advertisements
प्रश्न
When 0.4g of oxalic acid is dissolved in 40g of benzene, the freezing point of the solution is lowered by 0.45 K. Calculate the degree of association of acetic acid. Acetic acid forms dimer when dissolved in benzene.
(Kf for benzene = 5.12 K kg mol-1, at. wt. C = 12, H = 1, O = 16)
उत्तर
∆Tf = 0.45 K
i = ?
Kf = 5.12 K kg mol-1
MB of CH3COOH = 60
WA = 40 g
WB = 0.4 g
We know that
`triangle "T"_"f" = "i""K"_"f" . "W"_"B"/"M"_"B" xx 1000/"W"_"A"`
`0.45 = "i" xx 5.12 xx 0.4/60 xx 1000/40`
i = `(0.45 xx 60 xx 40)/(0.4 xx 1000 xx 5.12)`
`= (45 xx 60 xx 40)/(40 xx 1000 xx 5.12)`
`= 2700/5120 = 135/256 = 0.5273`
Let degree of association of acetic acid = α
∴ α = `("i" - 1)/(1/"n" - 1)` For CH3COOH , n = 2
`= (0.5273-1)/(1/2 -1) = 0.4727/0.5000 = 0.9514`
∴ Degree of association of acetic acid (α) = 0.9514 or 95.14%
APPEARS IN
संबंधित प्रश्न
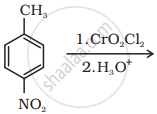
Write the structure of the product of the following reaction:
Ozonolysis of alkenes followed by the reaction with zinc dust and water gives ____________ depending on the substitution pattern of the alkene.
Esters react with DIBAL-H to produce:
The oxidation of toluene to benzoic acid can be done using which of the following reagents.
Can Gatterman-Koch reaction be considered similar to Friedel Craft’s acylation? Discuss.
An alkene ‘A’ (Mol. formula \[\ce{C5H10}\]) on ozonolysis gives a mixture of two compounds ‘B’ and ‘C’. Compound ‘B’ gives positive Fehling’s test and also forms iodoform on treatment with \[\ce{I2}\] and \[\ce{NaOH}\]. Compound ‘C’ does not give Fehling’s test but forms iodoform. Identify the compounds A, B and C. Write the reaction for ozonolysis and formation of iodoform from B and C.
Benz aldehyde + NaOH →
The strongest base among the following
When 2 – hydroxyl benzoic acid distilled with zinc dust, it give
The reaction of benzene with CO and HCl in the presence of anhydrous AlCl3 gives ______.
