Advertisements
Advertisements
प्रश्न
Can Gatterman-Koch reaction be considered similar to Friedel Craft’s acylation? Discuss.
उत्तर
Gattermann-Koch reaction is formylation (introduction of-CHO group) to the benzene nucleus. Friedel-Crafts acylation reaction is introduction of RCO-group in the benzene ring.


In Friedel-Crafts acylation reactions, the benzene is treated with an acid chloride in presence of anhydrous \[\ce{AlCl3}\]. So, Gattermann-Koch reaction can be considered similar to Friedel-Crafts acylation reaction.
APPEARS IN
संबंधित प्रश्न
Predict the product of the following reaction:
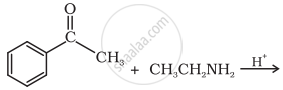
Name the electrophile produced in the reaction of benzene with benzoyl chloride in the presence of anhydrous \[\ce{AlCl3}\]. Name the reaction also.
An aromatic compound ‘A’ (Molecular formula \[\ce{C8H8O}\]) gives positive 2, 4-DNP test. It gives a yellow precipitate of compound ‘B’ on treatment with iodine and sodium hydroxide solution. Compound ‘A’ does not give Tollen’s or Fehling’s test. On drastic oxidation with potassium permanganate it forms a carboxylic acid ‘C’ (Molecular formula \[\ce{C7H6O2}\]), which is also formed along with the yellow compound in the above reaction. Identify A, B and C and write all the reactions involved.
What is the name of the given reaction of preparation of aldehyde?
\[\ce{C3COCl ->[H2][Pd/BaSO4] CH3CHO + HCl}\]
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Cl}
\end{array}\] obtained by chlorination of n-butane, will be
The strongest base among the following
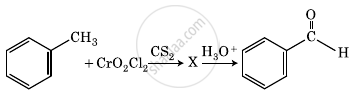
The intermediate compound ‘X’ in the following chemical reaction is:
Explain the following reactions:
Stephan reaction
An organic compound with molecular formula \[\ce{C7H7NO2}\] exists in three isomeric forms, the isomer ‘A’ has the highest melting point of the three. ‘A’ on reduction gives compound ‘B’ with molecular formula \[\ce{C7H9N}\]. ‘B’ on treatment with \[\ce{NaNO2/HCl}\] at 0-5° C to form compound ‘C’. On treating C with \[\ce{H3PO2}\], it gets converted to D with formula \[\ce{C7H8}\], which on further reaction with \[\ce{CrO2Cl2}\] followed by hydrolysis forms ‘E’ \[\ce{C7H6O}\]. Write the structure of compounds A to E. Write the chemical equations involved.
Account for the following:
Reduction of nitrobenzene using Fe and HCl is preferred over Sn and HCl.
