Advertisements
Advertisements
प्रश्न
Match the common names given in Column I with the IUPAC names given in Column II.
| Column I (Common names) |
Column II (IUPAC names) |
||
| (i) | Cinnamaldehyde | (a) | Pentanal |
| (ii) | Acetophenone | (b) | Prop-2-enal |
| (iii) | Valeraldehyde | (c) | 4-Methylpent-3-en-2-one |
| (iv) | Acrolein | (d) | 3-Phenylprop-2-enal |
| (v) | Mesityl oxide | (e) | 1-Phenylethanone |
उत्तर
| Column I (Common names) |
Column II (IUPAC names) |
||
| (i) | Cinnamaldehyde | (d) | 3-Phenylprop-2-enal |
| (ii) | Acetophenone | (e) | 1-Phenylethanone |
| (iii) | Valeraldehyde | (a) | Pentanal |
| (iv) | Acrolein | (b) | Prop-2-enal |
| (v) | Mesityl oxide | (c) | 4-Methylpent-3-en-2-one |
Explanation:
| (Common names) | Structure | (IUPAC names) |
| (i) Cinnamaldehyde | 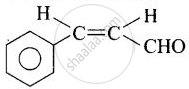 |
3-Phenylprop-2-enal |
| (ii) Acetophenone | 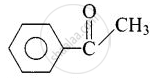 |
1-Phenylethanone |
| (iii) Valeraldehyde | 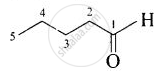 |
Pentanal |
| (iv) Acrolein | 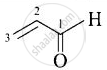 |
Prop-2-enal |
| (v) Mesityl oxide | 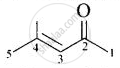 |
4-Methylpent-3-en-2- one |
APPEARS IN
संबंधित प्रश्न
Ozonolysis of alkenes followed by the reaction with zinc dust and water gives ____________ depending on the substitution pattern of the alkene.
Name the electrophile produced in the reaction of benzene with benzoyl chloride in the presence of anhydrous \[\ce{AlCl3}\]. Name the reaction also.
Match the example given in Column I with the name of the reaction in Column II.
| Column I (Example) |
Column II (Reaction) |
||
| (i) | \[\begin{array}{cc} \phantom{...}\ce{O}\phantom{..............................}\ce{O}\phantom{}\\ \phantom{...}||\phantom{..............................}||\phantom{}\\ \ce{CH3 - C - Cl + H2 ->[Pd - C/BasO4] CH3 - C - H} \end{array}\] |
(a) | Friedel Crafts acylation |
| (ii) | 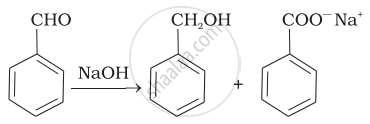 |
(b) | HVZ reaction |
| (iii) | 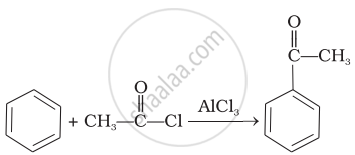 |
(c) | Aldol condensation |
| (iv) | \[\begin{array}{cc} \ce{R - CH2 - COOH ->[Br/Red P] R - CH - COOH}\\ \phantom{.....................}|\\ \phantom{.......................}\ce{Br} \end{array}\] |
(d) | Cannizaro’s reaction |
| (v) | \[\ce{CH3 - CN ->[(i) SnCl2/HCl][(ii) H2O/H+] CH3CHO}\] | (e) | Rosenmund’s reductio |
| (vi) | \[\ce{2CH3CHO ->[NaOH] CH3 - CH = CHCHO}\] | (f) | Stephen’s reaction |
An alkene ‘A’ (Mol. formula \[\ce{C5H10}\]) on ozonolysis gives a mixture of two compounds ‘B’ and ‘C’. Compound ‘B’ gives positive Fehling’s test and also forms iodoform on treatment with \[\ce{I2}\] and \[\ce{NaOH}\]. Compound ‘C’ does not give Fehling’s test but forms iodoform. Identify the compounds A, B and C. Write the reaction for ozonolysis and formation of iodoform from B and C.
What is the name of the given reaction of preparation of aldehyde?
\[\ce{C3COCl ->[H2][Pd/BaSO4] CH3CHO + HCl}\]
The reaction

The strongest base among the following
Aldehydes are the first oxidation products of ______.
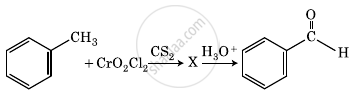
The intermediate compound ‘X’ in the following chemical reaction is:
Assertion (A): Strong oxidising agents oxidise toluene and its derivatives to benzoic acids.
Reason (R): It is possible to stop the oxidation of toluene at the aldehyde stage with suitable reagents.
Select the most appropriate answer from the options given below:
