Advertisements
Advertisements
प्रश्न
Which of the following reagents can be used to oxidise primary alcohols to aldehydes?
(i)
(ii)
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
उत्तर
(i)
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Explanation:
Strong oxidising agents such as acidified potassium permanganate are used for getting carboxylic acids from alcohols directly.
A better reagent for the oxidation of primary alcohols to aldehydes in good yield is pyridinium chlorochromate (PCC), a complex for chromium trioxide with pyridine and HCl.
APPEARS IN
संबंधित प्रश्न
Write IUPAC name of the following compound:
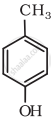
Write the structures of the products when Butan-2-ol reacts with CrO3
Write structural formulae for 1-Ethylcyclohexanol.
Write structural formulae for Cyclohex-2-en-1-ol.
The compound HOCH2 – CH2OH is __________.
Butane-2-ol is ____________.
Which of the following compounds will react with sodium hydroxide solution in water?
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: Phenols give o- and p-nitrophenol on nitration with conc.
Reason: –OH group in phenol is o–, p– directing.
Identify A and B in the following:

