Advertisements
Advertisements
प्रश्न
Why is respiration considered an exothermic reaction? Explain.
Explain why respiration is considered as an exothermic reaction. Give the chemical equation for this reaction.
उत्तर १
Respiration is considered as an exothermic reaction because, in respiration, the oxidation of glucose takes place, which produces large amount of heat energy.
\[\ce{C6H12O6(aq) + 6O2(g) -> 6CO2(g) + 6H2O(l) + energy}\]
उत्तर २
We get this energy from the food we eat. During digestion, food is broken down into simpler substances. For example, rice, potatoes and bread contain carbohydrates. These carbohydrates are broken down to form glucose. This glucose combines with oxygen in the cells of our body and provides energy. The special name of this reaction is respiration. Thus, respiration is an exothermic process because energy is produced during this process.
\[\ce{C6H12O6(aq) + 6O2(aq) -> 6CO2(aq) + 6H2O(l) + energy}\]
संबंधित प्रश्न
What happens chemically when quicklime is added to water filled in a bucket?
Gas A, which is the major cause of global warming, combines with hydrogen oxide B in nature in the presence of an environmental factor C and a green material D to form a six carbon organic compounds E and a gas F. The gas F is necessary for breathing.
(a) What is gas A?
(b) What is the common name of B?
(c) What do you think could be C?
(d) What is material D? Where is it found?
(e) Name the organic compound E.
(f) What is gas F? Name the natural process during which it is released.
what tpye of reaction is the following:
2K + Cl → 2KCl
Consider the following equation of the chemical reaction of a metal M:
4M + 3O2 → 2M2O3
This equation represents:
(a) combination reaction as well as reduction reaction
(b) decomposition reaction as well as oxidation reaction
(c) oxidation reaction as well as displacement reaction
(d) combination reaction as well as oxidation reaction
What happens during a chemical reaction ?
Fill in the blank
The chemical reaction between hydrogen and chlorine is a ................ reaction
A student prepared an aqueous solution of CuSO4 in beaker X and an aqueous solution of FeSO4 in beaker Y. He then dropped some iron pieces in beaker X and some zinc pieces in beaker Y. After about 10 hours he observed that the solutions in X and Y respectively appear:
(A) blue and green
(B) colourless and pale green
(C) colourless and light blue
(D) greenish and colourless
(a) What is double displacement reaction? Explain with an example.
(b) A small amount of quick lime is added to water in a beaker.
(i) Name and define the type of reaction that has taken place.
(ii) Write balanced chemical equation for the above reaction and the chemical name of the product formed.
(iii) List two main observations of this reaction.
Define: Chemical combination reaction
Classify the following reaction into different type:
CaO(s) + H2O(l) → Ca(OH)2(aq)
Give a balanced equation for –
A direct combination reaction involving two elements, one of which is a non-metal
H2(g) + Cl29(g) → 2HCl(g) is a ______
Define a combination reaction. Give one example of an exothermic combination reaction.
Which of the following are exothermic processes?
(i) Reaction of water with quick lime
(ii) Dilution of an acid
(iii) Evaporation of water
(iv) Sublimation of camphor (crystals)
The chemical reaction between copper and oxygen can be categorized as ______.
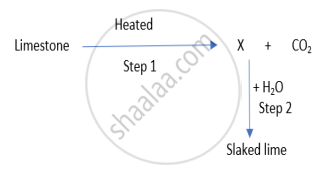
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
Give the chemical reaction when a few pieces of slaked lime are added to a beaker half-filled with water. Will it be an endothermic or exothermic reaction?
A clear solution of slaked lime is made by dissolving Ca(OH)2 in an excess of water. This solution is left exposed to air. The solution slowly goes milky as a faint white precipitate forms. Explain why a faint white precipitate forms, support your response with the help of a chemical equation.
A metal ribbon 'X' bums in oxygen with a dazzling white flame forming a white ash 'Y'. The correct description of X, Y and the type of reaction is:
