Advertisements
Advertisements
प्रश्न
88 cm3 of nitrogen is at a pressure of 770 mm mercury. If the pressure is raised to 880 mmHg, find by how much the volume will diminish, the temperature remaining constant.
उत्तर
V = 88 cm3
P = 770 mm
P1 = 880 mm
V1= ?
T = T1
Using gas equation,
`"PV"/"T" = ("P"_1 "V"_1)/"T"_1`
`(770 xx 88)/"T" = (880 xx "V"_1)/"T"`
V1 = 77 cm3
Volume diminishes = 88 - 77 = 11 cm3
APPEARS IN
संबंधित प्रश्न
State the law which is represented by the following graph:
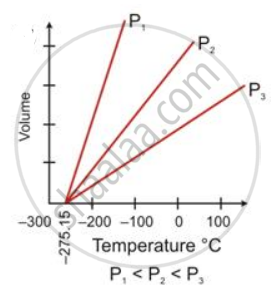
State the following:
Ice point in absolute temperature
Choose the correct answer:
The volume-temperature relationship is given by
It is found that on heating a gas its volume increases by 50% and its pressure decreases to 60% of its original value. If the original temperature was -15°C, find the temperature to which it was heated.
2500 cm3 of hydrogen is taken at STP. The pressure of this gas is further increased by two and a half times (temperature remaining constant). What volume will hydrogen occupy now?
Give its
(i) graphical representation,
(ii) mathematical expression
(iii) significance
Correct the following statement:
The volume of a fixed mass of a gas is directly proportional to its temperature, pressure remaining constant.
A certain mass of gas occupied 850 ml at a pressure of 760 mm of Hg. On increasing the pressure it was found that the volume of the gas was 75% of its initial value. Assuming constant temperature, find the final pressure of the gas?
It is required to reduce the volume of a gas by 20% by compressing it at a constant pressure. To do so, the gas has to be cooled. If the gas attains a final temperature of 157°C, find the initial temperature of the gas.
A fixed volume of a gas occupies 760cm3 at 27°C and 70 cm of Hg. What will be its vol. at s.t.p.
