Advertisements
Advertisements
प्रश्न
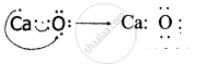
An atom X with atomic number 20 combines with atom Y with atomic number 8. Draw the dot structure for the formation of the molecule XY.
उत्तर
Atom X is Calcium with atomic number 20 and atom Y is Oxygen with atomic number 8.
APPEARS IN
संबंधित प्रश्न
Give electron dot diagram of the following:
nitrogen
What type of bond is formed between two atoms, when the electronegative difference between them is zero?
Element X is a metal with a valency 2; Y is a non-metal with a valency 3.
(a) Write an equation to show how Y forms as ion
(b) If Y is a diatomic gas, write an equation for the direct combination of X and Y to form a compound.
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2, 8, 1 |
2, 8, 7 |
2, 5 |
1 |
Answer the following question based on the table above:
What is the formula of the compound formed between W and X.
Why do `""_17^35"CI"` and `""_17^37"CI"` have the same chemical properties? In what respect do these atoms differ?
Explain
Duplet rule for the formation of hydrogen
Identify the following reaction as either oxidation or reduction:
O + 2e- → O2-
Complete the following table:
| Atoms | Atomic number | Electron distribution | |
| 1. | O | 8 | ______ |
| 2. | N | ______ | ______ |
| 3. | ______ | 17 | ______ |
| 4. | ______ | ______ | 2,8,2 |
The one which is composed of all the three kinds of bond.[ionic, covalent and coordinate bond]
Elements A, B and C have atomic number 17, 19 and 10 respectively.
State which one is a metal.
