Advertisements
Advertisements
प्रश्न
Assertion: \[\ce{Cu}\] cannot liberate hydrogen from acids.
Reason: Because it has positive electrode potential.
पर्याय
Both assertion and reason are true, and reason is the correct explanation of the assertion.
Both assertion and reason are true but reason is not the correct explanation of assertion.
Assertion is not true but reason is true.
Both assertion and reason are false.
उत्तर
Both assertion and reason are true, and reason is the correct explanation of the assertion.
Explanation:
\[\ce{Cu}\] lies below hydrogen in electrochemical series and has positive electrode potential. hence cannot liberate hydrogen from acids.
Both the statements assertion and reason are correct and reason is the correct explanation of assertion.
APPEARS IN
संबंधित प्रश्न
Account for the following:
Mn shows the highest oxidation state of +7 with oxygen but with fluorine, it shows oxidation state of +4.
Why are Mn2+ compounds more stable than Fe2+ towards oxidation to their +3 state?
How would you account for the following?
Transition metals and their compounds act as catalysts.
Read the passage given below and answer the following question:
The transition metals when exposed to oxygen at low and intermediate temperatures form thin, protective oxide films of up to some thousands of Angstroms in thickness. Transition metal oxides lie between the extremes of ionic and covalent binary compounds formed by elements from the left or right side of the periodic table. They range from metallic to semiconducting and deviate by both large and small degrees from stoichiometry. Since electron bonding levels are involved, the cations exist in various valence states and hence give rise to a large number of oxides. The crystal structures are often classified by considering a cubic or hexagonal close-packed lattice of one set of ions with the other set of ions filling the octahedral or tetrahedral interstices. The actual oxide structures, however, generally show departures from such regular arrays due in part to distortions caused by packing of ions of different size and to ligand field effects. These distortions depend not only on the number of d-electrons but also on the valence and the position of the transition metal in a period or group.
In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage.
Assertion: Cations of transition elements occur in various valence states.
Reason: Large number of oxides of transition elements are possible.
Read the passage given below and answer the following question:
The transition metals when exposed to oxygen at low and intermediate temperatures form thin, protective oxide films of up to some thousands of Angstroms in thickness. Transition metal oxides lie between the extremes of ionic and covalent binary compounds formed by elements from the left or right side of the periodic table. They range from metallic to semiconducting and deviate by both large and small degrees from stoichiometry. Since electron bonding levels are involved, the cations exist in various valence states and hence give rise to a large number of oxides. The crystal structures are often classified by considering a cubic or hexagonal close-packed lattice of one set of ions with the other set of ions filling the octahedral or tetrahedral interstices. The actual oxide structures, however, generally show departures from such regular arrays due in part to distortions caused by packing of ions of different size and to ligand field effects. These distortions depend not only on the number of d-electrons but also on the valence and the position of the transition metal in a period or group.
In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage.
Assertion: Crystal structure of oxides of transition metals often show defects.
Reason: Ligand field effect cause distortions in crystal structures.
It has been observed that first ionization energy of 5 d series of transition elements are higher than that of 3d and 4d series, explain why?
Which of the following ions will exhibit colour in aqueous solution?
Give reason for the following statement:
Physical and chemical properties of the 4d and 5d series of the transition elements are quite similar to expected.
The value of Δ0 for \[\ce{RhCl^{3-}6}\] is 243 KJ/mol which wavelength of light will promote an electron from. The colour of the complex is ______.
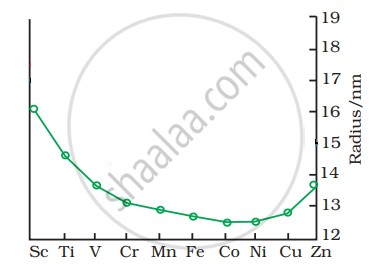
The trend of which property is represented by the following graph?