Advertisements
Advertisements
प्रश्न
Consider a rectangular block of wood moving with a velocity v0 in a gas at temperature T and mass density ρ. Assume the velocity is along x-axis and the area of cross-section of the block perpendicular to v0 is A. Show that the drag force on the block is `4ρAv_0 sqrt((KT)/m)`, where m is the mass of the gas molecule.
उत्तर
Consider the diagram
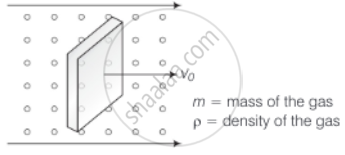
Let n = number of molecules per unit volume
Vrms = rms speed of the gas molecules
When a block is moving with speed v0, relative speed of molecules w.r.t. front face = v + v0
Coming head-on, momentum transferred to block per collision = 2m (v + v0), where, m = mass of the molecule.
The number of collisions in time Δt = m(v + v0)2nAΔt, where, A = area of cross-section of block and factor of 1/2 appears due to particles moving towards the block.
∴ The momentum transferred in time Δt = m(v + v0)2nAΔt from the front surface.
Similarly, momentum transferred in time Δt = m(v – v0)2nAΔt ......(From the back surface)
∴ Net force (drag force) = mnA[(v + v0)2 – (v – v0)2] .....[From front]
= mnA (4vv0)
= (4 mnAv)v0
= (4ρAv)v0 ......(i)
Where we have assumed ρ = `(mn)/V = M/V`
If v = velocity along the x-axis
Then, we can write KE = `1/2 mv^2 = 1/2 K_BT`
⇒ `v = sqrt((K_BT)/m)` .....`[(K_B = "Boltzmann constant"),(KE = "Kinetic energy"),(T = "Temperature")]`
∴ From equation (i), Drag force = (4ρAv)v0 = `4ρA sqrt((K_BT)/m) v_0`.
APPEARS IN
संबंधित प्रश्न
Consider a gas of neutrons. Do you expect it to behave much better as an ideal gas as compared to hydrogen gas at the same pressure and temperature?
0.040 g of He is kept in a closed container initially at 100.0°C. The container is now heated. Neglecting the expansion of the container, calculate the temperature at which the internal energy is increased by 12 J.
Use R = 8.3 J K-1 mol-1
An ideal gas is kept in a long cylindrical vessel fitted with a frictionless piston of cross-sectional area 10 cm2 and weight 1 kg in figure. The vessel itself is kept in a big chamber containing air at atmospheric pressure 100 kPa. The length of the gas column is 20 cm. If the chamber is now completely evacuated by an exhaust pump, what will be the length of the gas column? Assume the temperature to remain constant throughout the process.

The temperature and the dew point in an open room are 20°C and 10°C. If the room temperature drops to 15°C, what will be the new dew point?
An adiabatic cylindrical tube of cross-sectional area 1 cm2 is closed at one end and fitted with a piston at the other end. The tube contains 0.03 g of an ideal gas. At 1 atm pressure and at the temperature of the surrounding, the length of the gas column is 40 cm. The piston is suddenly pulled out to double the length of the column. The pressure of the gas falls to 0.355 atm. Find the speed of sound in the gas at atmospheric temperature.
Compare the rate of radiation of metal bodies at 727 °C and 227 °C.
Average kinetic energy of H2 molecule at 300K is 'E'. At the same temperature, average kinetic energy of O2 molecule will be ______.
An insulated container containing monoatomic gas of molar mass m is moving with a velocity vo. If the container is suddenly stopped, find the change in temperature.
Explain why there is no atmosphere on moon.
A proton, a deuteron and an α-particle with same kinetic energy enter into a uniform magnetic field at right angle to magnetic field. The ratio of the radii of their respective circular paths is ______.
