Advertisements
Advertisements
प्रश्न
Distinguish between Electrophile and nucleophile.
उत्तर
| No. | Electrophile | Nucleophile |
| 1. | An electrophile is an electron-deficient species. | A nucleophile is an electron-rich species. |
| 2. | It is attracted towards negative charges (electron seeking). | It is attracted towards a positive charge (nucleus seeking). |
| 3. | It attacks a nucleophilic center in the substrate and brings about an electrophilic reaction | It attacks the electrophilic center in the substrate and brings about a nucleophilic reaction. |
| 4. | It is an electron pair acceptor. (Lewis acid) | It is an electron-pair donor. (Lewis base) |
| 5. | It can be a positively charged ion or a neutral species having a vacant orbital. | It can be negatively charged ion or neutral species having at least one lone pair of electrons |
| 6. | e.g. H+, Br+,\[\ce{NO^+_2}\], BF3, AlCl3, etc. | e.g. OH-, Cl-, CN-, etc. |
APPEARS IN
संबंधित प्रश्न
Find out the most stable species from the following. Justify.
`bar"C""H"_3, bar"C""H"_2"Br", bar"C""Br"_3`
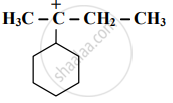
Identify the α - carbons in the following species and give the total number of α-hydrogen.
Draw a resonance structure of the following:
Benzaldehyde
Distinguish between Inductive effect and resonance effect.
Distinguish between Homolysis and heterolysis.
Write true or false. Correct the false statement.
Aniline is a heterocyclic compound.
Draw all the no-bond resonance structures of isopropyl carbocation.
Choose the correct option.
Which of the following statements are true with respect to electronic displacement in a covalent bond?
a. Inductive effect operates through π bond
b. Resonance effect operates through σ bond
c. Inductive effect operates through σ bond
d. Resonance effect operates through π bond
Choose the correct option.
The geometry of a carbocation is ______.
Choose the correct option.
The homologous series of alcohols has general molecular formula ______.
Which of the following is the strongest nucleophile?
Which of the following alkyl groups shows maximum positive inductive effect?
The overlap of σ-p orbitals is called ____________.
How many pi bonds and sigma bonds are present in following molecule?
Which of the following statements is not correct?
Identify the group that exerts electron withdrawing resonance effect.
Identify the functional group that has an electron-donating inductive effect.
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogens.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
