Advertisements
Advertisements
प्रश्न
Explain a process in which a biocatalyst is used in industrial preparation of a compound known to you.
उत्तर
Ethanol, \[\ce{C2H5OH}\], is obtained commercially by fermentation, the oldest method is from sugars. The sugar in molasses, sugarcane or fruits such as grapes is converted to glucose and fructose, (both of which have the formula \[\ce{C6H12O6}\]), in the presence of an enzyme, invertase. Glucose and fructose undergo fermentation in the presence of another enzyme, zymase, which is found in yeast.
\[\ce{C12H22O11 + H2O ->[Invertase] \underset{Glucose}{C6H12O6} + \underset{Fructose}{C6H12O6}}\]
\[\ce{C6H12O6 ->[Zymase] 2C2H5OH + 2CO2}\]
In wine making, grapes are the source of sugars and yeast. As grapes ripen, the quantity of sugar increases and yeast grows on the outer skin. When grapes are crushed, sugar and the enzyme come in contact and fermentation starts. Fermentation takes place in anaerobic conditions i.e. in absence of air. Carbon dioxide is released during fermentation.
APPEARS IN
संबंधित प्रश्न
Predict the major product of acid catalysed dehydration of butan-1-ol.
Show how you would synthesise the following alcohol from an appropriate alkene?
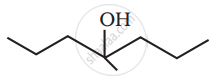
Write the structures of A, B and C in the following reactions :
Commercially carboxylic acids are reduced to alcohols by converting them to the ______.
How can propan-2-one be converted into tert- butyl alcohol?
Magnesium wire continues to burn in the atmosphere of CO2 because
Most readily hydrolysed halide is:-
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.

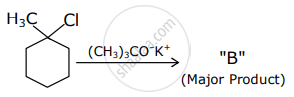
The products "A" and "B" formed in above reactions are:
Write the mechanism of acid dehydration of ethanol to yield ethene.
