Advertisements
Advertisements
प्रश्न
Give IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{.......}\ce{CH3}
\end{array}\]
उत्तर
1-Ethoxy-2-methylpropane
APPEARS IN
संबंधित प्रश्न
What is metamerism?
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
\phantom{}|\phantom{.............}|\phantom{......}|\phantom{.........}\\
\phantom{}\ce{OH}\phantom{..........}\ce{OH}\phantom{...}\ce{C2H5}\phantom{......}
\end{array}\]
Write IUPAC name of the following compound:
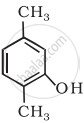
Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
Write the structure and IUPAC name of 'methyl-n-propyl ether'.
3-Methylbutane-2-ol on heating with HI gives ______
In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
One of the following is not a dihydroxy derivative of benzene.
An example of a compound with functional group – O – is ____________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
The IUPAC name of the ether CH2 = CH–CH2OCH3 is:
Among the following sets of reactants which one produces anisole?
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Write the IUPAC name of the following compound.
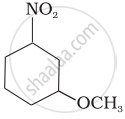
Write chemical reactions for the following conversion:
Acetic acid into ethyl alcohol
The IUPAC name of 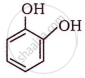 is ______.
is ______.
Write the IUPAC name.
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C -CH3}\\
\phantom{.}|\phantom{......}|\phantom{......}|\\
\phantom{....}\ce{CH3\phantom{...}\ce{OH}\phantom{...}\ce{CH3}}\
\end{array}\]
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
