Advertisements
Advertisements
प्रश्न
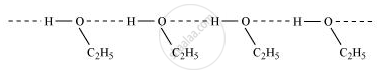
Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
उत्तर
Ethanol undergoes intermolecular H-bonding due to the presence of −OH group, resulting in the association of molecules. Extra energy is required to break these hydrogen bonds. On the other hand, methoxymethane does not undergo H-bonding. Therefore, ethanol has a higher boiling point than methoxymethane.
APPEARS IN
संबंधित प्रश्न
Give reasons for the following: Butan-1-ol has a higher boiling point than diethyl ether.
Give reasons for the following : Phenol is more acidic than ethanol.
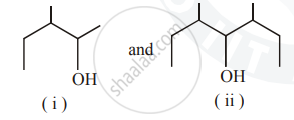
Identify the chiral molecule in the following pair :
Write the structures of A, B and C in the following reactions :
\[ C_6 H_5 {NO}_2 \to^{Sn/HCI} A \to^{{NaNO}_2 /HCI}_{273 K} B \to^{H_2 O}_∆ C\]
Account for the following:
CH3CHO is more reactive than CH3COCH3 towards reaction with HCN.
Write a chemical reaction to show that the open structure of D-glucose contains the following :
Five alcohol groups
Isopropyl alcohol is obtained by reacting which of the following alkenes with concentrated H2SO4 followed by boiling with H2O?
Alcohols of low molecular weight are _____________.
Arrange the following compounds in increasing order of boiling point. Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol
A solution of phenol in chloroform when treated with aqueous NaOH gives compound P as a major product. The mass percentage of carbon in P is ______. (to the nearest integer) (Atomic mass: C = 12; H = 1; O = 16)
Arrange the following in order of increasing boiling point:
Ethoxyethane, Butanal, Butanol, n-butane
Assertion (A): Alcohols react both as nucleophiles and electrophiles.
Reason (R): The bond between C–O is broken when alcohols react as nucleophiles.
Select the most appropriate answer from the options given below:
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
How is the following conversion carried out?
Methyl magnesium bromide → 2-Methylpropan-2-ol.
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide-> 2-Methylpropan-2-ol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
How is the following conversions carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\].
