Advertisements
Advertisements
प्रश्न
How will you convert ethanal into the following compound?
Butane-1, 3-diol
उत्तर
\[\ce{\underset{Ethanal}{2CH3CHO}->[Dil.NaOH][Aldol condensation]\underset{3-Hydroxybutanal}{\overset{4}{C}H3\overset{3}{C}HOH - \overset{2}{C}H2 - \overset{1}{C}HO}->[NaBH4][(Reduction)]\underset{Butane-1, 3-diol}{\overset{4}{C}H3 - \overset{3}{C}HOH - \overset{2}{C}H2 - \overset{1}{C}H2OH}}\]
APPEARS IN
संबंधित प्रश्न
A compound 'A' of molecular formula C2H3OCl undergoes a series of reactions as shown below. Write the structures of A, B, C and D in the following reactions :
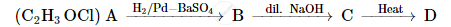
How will you bring about the following conversion?
Ethanal to but-2-enal
Which of the following compounds would undergo aldol condensation, which the Cannizzaro reaction and which neither? Write the structures of the expected products of aldol condensation and Cannizzaro reaction.
- Methanal
- 2-Methylpentanal
- Benzaldehyde
- Benzophenone
- Cyclohexanone
- 1-Phenylpropanone
- Phenylacetaldehyde
- Butan-1-ol
- 2, 2-Dimethylbutanal
How will you bring about the following conversion in not more than two steps?
Ethanol to 3-Hydroxybutanal
How will you bring about the following conversion in not more than two steps?
Benzaldehyde to 3-Phenylpropan-1-ol
Write chemical equations of the following reaction :
Benzoyl chloride is hydrogenated in the presence of `"Pd"/(BaSO_4)`
Explain aldol condensation reaction in detail.
Which of the following compounds do not undergo aldol condensation?
(i) \[\ce{CH3 - CHO}\]
(ii) 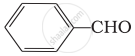
(iii) \[\begin{array}{cc}
\phantom{}\ce{O}\\
\phantom{}||\\
\ce{CH3 - C - CH3}
\end{array}\]
(iv) \[\begin{array}{cc}
\phantom{}\ce{CH3}\\
|\phantom{...}\\
\ce{CH3 - C - CHO}\phantom{..}\\
|\phantom{...}\\
\phantom{}\ce{CH3}\\
\end{array}\]
Why is there a large difference in the boiling points of butanal and butan-1-ol?
What product will be formed on reaction of propanal with 2-methylpropanal in the presence of \[\ce{NaOH}\]? What products will be formed? Write the name of the reaction also.
Cross aldol condensation occurs between
\[\ce{CH3-CH2-CHO ->[dil][alkali] Product}\]
The product in the above reaction is:
Explain Aldol condensation of ethanal.
Which of the following does not give aldol condensation reaction?
Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
Write a note on the aldol condensation reaction of acetaldehyde.
