Advertisements
Advertisements
प्रश्न
How would you account for the following?
Transition metals exhibit variable oxidation states.
उत्तर
(i) The variable oxidation states of transition elements are due to the participation of ns and (n−1)d-electrons in bonding. Lower oxidation state is exhibited when ns-electrons take part in bonding. Higher oxidation states are exhibited when (n − 1) d-electrons take part in bonding.
APPEARS IN
संबंधित प्रश्न
How would you account for the following: Transition metals form complex compounds.
Calculate the ‘spin only’ magnetic moment of \[\ce{M^{2+}_{ (aq)}}\] ion (Z = 27).
To what extent do the electronic configurations decide the stability of oxidation states in the first series of the transition elements? Illustrate your answer with examples.
Compare the stability of +2 oxidation state for the elements of the first transition series.
What are alloys?
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Electronic configurations
Give reasons Iron has the higher enthalpy of atomization than that of copper.
Account for the following:
In case of transition elements, ions of the same charge in a given series show progressive decrease in radius with increasing atomic number.
Which does not belong to first transition series?
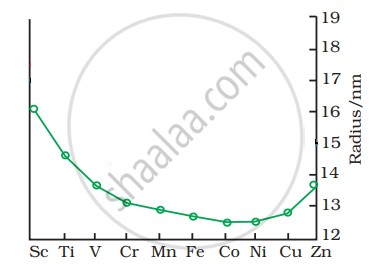
The trend of which property is represented by the following graph?