Advertisements
Advertisements
प्रश्न
How would you account for the following:
The d1 configuration is very unstable in ions.
उत्तर
In ions, the d1 configuration is unstable, but after losing one electron, it becomes more stable due to the vacant d-orbitals. All elements with d1 configuration are reduced or undergo disproportionation. e.g.,
\[\ce{3\overset{+6}{\underset{3d^1}{Mn}}O^2-_4 + 4H+ -> 2\overset{+7}{\underset{3d^0}{Mn}}O^-_4 + \overset{+4}{Mn}O2 + 2H2O}\]
APPEARS IN
संबंधित प्रश्न
Calculate magnetic moment of `Fe_((aq))^(2+) ion (Z=26).`
Account for the following:
Mn shows the highest oxidation state of +7 with oxygen but with fluorine, it shows oxidation state of +4.
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Electronic configurations
Write down the number of 3d electrons in the following ion:
Cu2+
Indicate how would you expect the five 3d orbitals to be occupied for this hydrated ions (octahedral).
What can be inferred from the magnetic moment value of the following complex species?
| Example | Magnetic Moment (BM) |
| K2[MnCl4] | 5.9 |
Explain why Zn2+ salts are white whereas Cu2+ salts are coloured.
Transition metals with highest melting point is ____________.
\[\ce{KMnO4}\] acts as an oxidising agent in acidic medium. The number of moles of \[\ce{KMnO4}\] that will be needed to react with one mole of sulphide ions in acidic solution is ______.
Which of the following ions show higher spin only magnetic moment value?
(i) \[\ce{Ti^3+}\]
(ii) \[\ce{Mn2+}\]
(iii) \[\ce{Fe2+}\]
(iv) \[\ce{Co3+}\]
Why EΘ values for Mn, Ni and Zn are more negative than expected?
Assertion: \[\ce{Cu^2+}\] iodide is not known.
Reason: \[\ce{Cu^2+}\] oxidises \[\ce{I^-}\] to iodine.
Identify A to E and also explain the reactions involved.
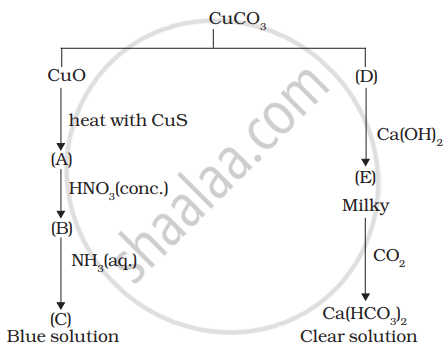
Answer the following question:
Which element of the first transition series has highest second ionisation enthalpy?
Mention any three processes where transition metals act as catalysts.
On the basis of the figure given below, answer the following questions:

- Why Manganese has lower melting point than Chromium?
- Why do transition metals of 3d series have lower melting points as compared to 4d series?
- In the third transition series, identify and name the metal with the highest melting point.
A metallic ion 'M' reacts with chloride ion to form white precipitate which is readily soluble in aqueous ammonia. Identify 'M'?
Why Zn, Cd and Hg are not called transition metals?
In the ground state of atomic Fe (Z = 26), the spin-only magnetic moment is ______ × 10-1 BM.
(Round off to the nearest integer).
[Given: `sqrt3 = 1.73, sqrt2 = 1.41`]
A pair of coloured ions is ______.
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Oxidation states
