Advertisements
Advertisements
प्रश्न
Give the formula and describe the structure of a noble gas species which is isostructural with
`ICI_4^(-)`
उत्तर १
XeF4 is isoelectronic with `ICI_4^(-)` and has square planar geometry.
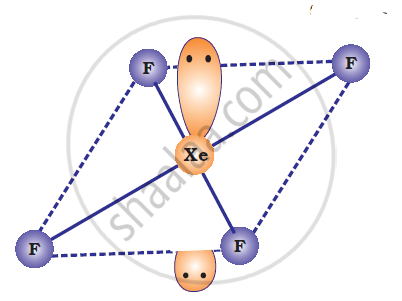
उत्तर २
ICI4–: In ICI4–, central atom I has seven valence electrons and one due to negative charge. Four out of these 8 electrons are utilized in forming four single bonds with four Cl atoms. Four remaining electrons constitutes the two lone pairs. It is arranged in square planar structure. ICI4– has 36 valence electrons. A noble gas species having 36 valence electrons is XeF4 (8 + 4 x 7 = 36). XeF4 is also square planar.
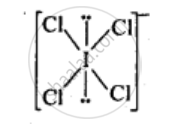
APPEARS IN
संबंधित प्रश्न
Account for the following:
Helium is used in diving apparatus.
What happens when: XeF4 reacts with SbF5?
Complete the following equations : XeF4 + O2F2→
What inspired N. Bartlett for carrying out reaction between Xe and PtF6?
How are XeO3 and XeOF4 prepared?
Answer the following.
List the uses of Neon and argon gases.
Complete the following reactions : XeF6 + 3H2O →
Complete the following reactions:
XeF6+2H2O ----->
Draw the structures of the following
XeF6
Draw the structure of XeF4.
Sulfur dioxide reacts with sodium hydroxide solution to form _______.
Partial hydrolysis of XeF4 gives ____________.
Which of the following reactions is an example of redox reaction?
On partial hydrolysis, XeF6 gives ______.
Noble gases are named because of their inertness towards reactivity. Identify an incorrect statement about them.
\[\ce{XeF4 + H2O - \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
