Advertisements
Advertisements
प्रश्न
Propanoic acid to ethylamine.
उत्तर
\[\ce{\underset{\text{Propanoic acid}}{CH_3CH_2COOH} + NH_3 -> CH_3 CH_2COONH_4 ->[{Δ}][{-H_2O}] CH_3 CH_2CONH_2 ->[{Br_2+4NaOH}][{Δ}]\underset{\text{Ethyl amine}}{CH_3CH_2NH_2}+ {Na_2 CO_3} + {2NaBr +2H_2 O}}\]
APPEARS IN
संबंधित प्रश्न
Name the following compound according to IUPAC system.
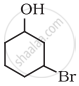
Write IUPAC name of the following compound:
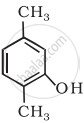
Write IUPAC name of the following compound:
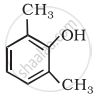
Give IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
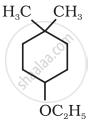
Give IUPAC name of the following ether:
Which of the following compounds is NOT prepared by the action of alcoholic NI3 on alkyl halide?
(a) CH3NH2
(b) CH3- CH2- NH2
(c) CH3 - CH2 - CH2 - NH2
(d) (CH3)3 C- NH2
Write the structures of the products when Butan-2-ol reacts with CrO3
Write the IUPAC name of the following :
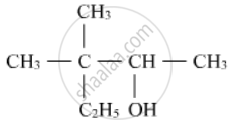
Write IUPAC names of the following
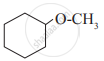
Glycerol is ____________.
HBr reacts fastest with ____________.
IUPAC name of the compound is:
\[\begin{array}{cc}
\ce{CH3-CH-OCH3}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\]
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Assertion: Phenols give o- and p-nitrophenol on nitration with conc. \[\ce{HNO3}\] and \[\ce{H2SO4}\] mixture.
Reason: –OH group in phenol is o–, p– directing.
Write chemical reactions for the following conversion:
Acetic acid into ethyl alcohol
Identify A and B in the following:

How are the following conversions carried out?
Methyl magnesium bromide→2-Methylpropan-2-ol.
Write structural formulae for:
p-Nitrophenol
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.....}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{.}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
