Advertisements
Advertisements
Question
Propanoic acid to ethylamine.
Solution
\[\ce{\underset{\text{Propanoic acid}}{CH_3CH_2COOH} + NH_3 -> CH_3 CH_2COONH_4 ->[{Δ}][{-H_2O}] CH_3 CH_2CONH_2 ->[{Br_2+4NaOH}][{Δ}]\underset{\text{Ethyl amine}}{CH_3CH_2NH_2}+ {Na_2 CO_3} + {2NaBr +2H_2 O}}\]
APPEARS IN
RELATED QUESTIONS
What is metamerism?
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\phantom{........................}\ce{CH2OH}\\
\phantom{..................}|\\
\ce{CH3 - CH - CH2 - CH - CH - CH3}\\
\phantom{}|\phantom{.............}|\phantom{........}\\
\phantom{..}\ce{CH3}\phantom{..........}\ce{OH}\phantom{........}
\end{array}\]
Write IUPAC name of the following compound:
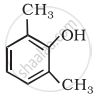
Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
Give IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
3-Methylbutane-2-ol on heating with HI gives ______
C6H5OCH2CH3 is called:
An example of a compound with functional group – O – is ____________.
The correct acidic strength order of the following is:
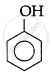
(I)

(II)
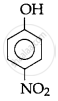
(III)
IUPAC name of m-cresol is ______.
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
Explain why p-nitrophenol is more acidic than phenol.
Explain why Lewis acid is not required in bromination of phenol?
How can phenol be converted to aspirin?
Convert the following:
Ethyl alcohol into ethyl acetate
Write chemical reactions for the following conversion:
Acetic acid into ethyl alcohol
The IUPAC name of 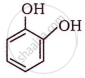 is ______.
is ______.
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{...}OH\phantom{...}CH3}\\
\end{array}\]
