Advertisements
Advertisements
प्रश्न
The energy levels of an atom of a certain element are shown in the given figure. Which one of the transitions A, B, C, D or E will result in the emission of photons of electromagnetic radiation of wavelength 618.75 nm? Support your answer with mathematical calculations.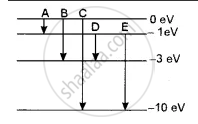
उत्तर
The wavelength emitted λ = 618.75 nm
∴ Corresponding energy E = h `c/λ`
= `( 6.6 xx 10^-34 xx 3 xx 10^8 )/( 618.75 xx 10^-9 xx 1.6 xx 10^-19) e^V`
= `( 6.6 xx 3 )/( 618.75 xx 1.6 ) xx 100`
= 2eV
This corresponds to the transition D.
In which energy emitted is ΔE = - 1 - ( - 3 ) = 2eV
APPEARS IN
संबंधित प्रश्न
How are electromagnetic waves produced?
Suppose that the electric field amplitude of an electromagnetic wave is E0 = 120 N/C and that its frequency is v = 50.0 MHz.
- Determine B0, ω, k, and λ.
- Find expressions for E and B.
Suppose that the electric field part of an electromagnetic wave in vacuum is
`vec"E" = {(3.1"N"/"C") cos[(1.8 ("rad")/"m")"y" + (5.4 xx 10^8 ("rad")/"s")"t"]}hat"i"`
(a) What is the direction of propagation?
(b) What is the wavelength λ?
(c) What is the frequency v?
(d) What is the amplitude of the magnetic field part of the wave?
(e) Write an expression for the magnetic field part of the wave.
Which of the following have zero average value in a plane electromagnetic wave?
(a) Electric field
(b) Magnetic field
(c) Electric energy
(d) Magnetic energy
Let E = E0 sin[106 x -ωt] be the electric field of plane electromagnetic wave, the value of ω is ______.
Write a short note on the visible spectrum.
Velocity of plane electromagnetic waves in vacuum equals ______.
Which of the following electromagnetic radiations has the smallest wave length?
Which of the following are not electromagnetic waves?
Why is the orientation of the portable radio with respect to broadcasting station important?
