Advertisements
Advertisements
प्रश्न
Using de Broglie’s hypothesis, explain with the help of a suitable diagram, Bohr’s second postulate of quantization of energy levels in a hydrogen atom.
उत्तर
De Broglie’s Explanation of Bohr’s Second Postulate of Quantisation
• De-Broglie’s hypothesis that electron has a wavelength λ = h/mv gave an explanation for Bohr’s quantised orbits by bringing in the wave particle duality.
• Orbits correspond to circular standing waves in which the circumference of the orbits equal whole number of wavelength.
According to de Broglie’s hypothesis
`lambda = h/p`also for Bohr’s model
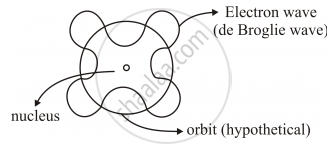
It states,
`nlambda = 2pir`
`=> n h/p = 2pir`
`=> rp =n h/(2pi) ` (as rp =L)
`=> L=n h/(2pi)` Bohr's 2ndPostulate
APPEARS IN
संबंधित प्रश्न
A proton and an α-particle are accelerated through the same potential difference. Which one of the two has less kinetic energy? Justify your answer.
State how de-Broglie wavelength (`lambda`) of moving particles varies with their linear momentum (p).
Answer the following question.
Obtain the expression for the ratio of the de-Broglie wavelengths associated with the electron orbiting in the second and third excited states of the hydrogen atom.
A litre of an ideal gas at 27°C is heated at constant pressure to 297°C. The approximate final volume of the gas is?
A solid sphere is in a rolling motion. In rolling motion, a body possesses translational kinetic energy (kt) as well as rotational kinetic energy (kr) simultaneously. The ratio kt : kr for the sphere is:
If the kinetic energy of the particle is increased to 16 times its previous value, the percentage change in the de-Broglie wavelength of the particle is:
When the displacement of a particle executing simple harmonic motion is half of its amplitude, the ratio of its kinetic energy to potential energy is:
A proton is accelerated through one volt the increase in its kinetic energy is approximately
The kinetic energy of electron in (electron volt) moving with the velocity of 4 × 106 m/s will be
Number of ejected photo electrons increase with increase
