Advertisements
Advertisements
प्रश्न
What are the limitations of the first law of thermodynamics?
उत्तर
Limitations of the first law of thermodynamics: The first law of thermodynamics explains well the inter convertibility of heat and work. But it does not indicate the direction of change.
For example,
(a) When a hot object is in contact with a cold object, heat always flows from the hot object to the cold object but not in the reverse direction. According to the first law, it is possible for the energy to flow from a hot object to a cold object or from a cold object to a hot object. But in nature, the direction of heat flow is always from higher temperature to lower temperature.
(b) When brakes are applied, a car stops due to friction and the work done against friction is converted into heat. But this heat is not reconverted to the kinetic energy of the car. So the first law is not sufficient to explain many natural phenomena.
APPEARS IN
संबंधित प्रश्न
Give an example of some familiar process in which heat is added to an object, without changing its temperature.
Draw a p-V diagram showing positive work at constant pressure.
Differentiate between the reversible and irreversible processes.
Explain graphically (i) positive work with varying pressure, (ii) negative work with varying pressure, and (iii) positive work at constant pressure.
In an isochoric process, we have ____________.
Apply first law for an adiabatic process.
What is a cyclic process?
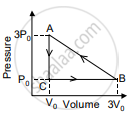
An ideal gas is taken in a cyclic process as shown in the figure. Calculate
- work done by the gas
- work done on the gas
- Net work done in the process

In which of the following processes, beat is neither absorbed nor released by a system?
In the figure shown here, the work done in the process ACBA is ______.