Advertisements
Advertisements
प्रश्न
Explain in detail the isochoric process.
उत्तर
Isochoric process: This is a thermodynamic process in which the volume of the system is kept constant. But pressure, temperature and internal energy continue to be variables.
The pressure-volume graph for an isochoric process is a vertical line parallel to the pressure axis as shown in Figure.

Isochoric process with increased pressure
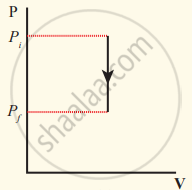
Isochoric process with decreased pressure

Isochoric process
P = `((μ"R")/"V")"T"` ...............(1)
where `((μ"R")/"V")` = constant
We can infer that the pressure is directly proportional to temperature. This implies that the P-T graph for an isochoric process is a straight line passing through the origin. If gas goes from the state (Pi, Ti) to (Pf, Tf) at constant volume, then the system satisfies the following equation
`"P"_"i"/"T"_"i" = "P"_"f"/"T"_"f"` .....,,,(2)
For an isochoric processes, ∆V = 0 and W = 0. Then the first law becomes
∆U = 0 .................(3)
Implying that the heat supplied is used to increase only the internal energy. As a result the temperature increases and pressure also increases.
Suppose a system loses heat to the surroundings through conducting walls by keeping the volume constant, then its internal energy decreases. As a result the temperature decreases; the pressure also decreases.
APPEARS IN
संबंधित प्रश्न
For work done to be reversible, the process should be ______
Draw a p-V diagram showing positive work with varying pressure.
Explain the thermodynamics of the isobaric process.
Explain thermodynamics of the adiabatic process.
The V-T diagram of an ideal gas which goes through a reversible cycle A→B→C→D is shown below. (Processes D→A and B→C are adiabatic)
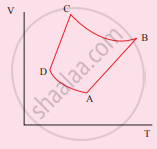
The corresponding PV diagram for the process is (all figures are schematic)
Apply first law for an adiabatic process.
Apply first law for an isobaric process.
For a given ideal gas 6 × 105 J heat energy is supplied and the volume of gas is increased from 4 m3 to 6 m3 at atmospheric pressure. Calculate
- the work done by the gas
- change in internal energy of the gas
- graph this process in PV and TV diagram
Assertion: Equal volumes of monatomic and polyatomic gases are adiabatically compressed separately to equal compression ratio `("P"_2/"P"_1)`. Then monatomic gas will have greater final volume.
Reason: Among ideal gases, molecules of a monatomic gas have the smallest number of degrees of freedom.
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
