Advertisements
Advertisements
प्रश्न
What observation you will record and how will you determine the specific latent heat of fusion of ice?
उत्तर
A clean and dry copper calorimeter with a strirrer, is weighed when empty, again weighed when filled nearly 2/3 with water. Then initial temperature is noted. Some pieces of dry ice are transferred into the calorimeter. The calorimeter is placed inside the box and is continuously stirred till the whole of ice is melted. The final temperature is carefully noted. Then the mass of calorimeter with water and ice is again weighed.
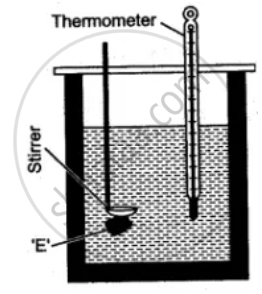
The observations recorded are:
Mass of empty calorimeter with stirrer = m1 gm.
Mass of calorimeter + water = m2 gm.
Initial temperature of water = t1 °C
Final temperature of mixture = t2 °C
Mass of calorimeter + Water + Ice (in the form of water) = m3 gm.
Temperature of ice = 0 °C
If L j/gm be the specific latent heat of fusion of ice and sc be the specific heat capacity of copper, then:
Mass of ice method = (m3 - m1) gm
Heat required by ice water at 0 °C to rise to t2 °C
= (m3 - m2) × 4.2 × (t2 - 0)
Heat lost by calorimeter = m1 × Sc × (t1 - t2)
Heat lost by water = (m2 - m1) × 4.2 × (t1 - t2)
From the heat equation,
Heat gained = Heat lost
(m3 - m2) L + (m3 - m2) × 4.2 × t2 = m1 × sc (t1 - t2) + (m2 - m1) × 4.2 × (t1 - t2)
Therefore,
L = `[(("m"_1"s"_"c")("t"_1 - "t"_2) + ("m"_2 - "m"_1) xx 4.2 xx ("t"_1 - "t"_2) - ("m"_3 - "m"_2) xx 4.2 xx "t"_2)/(("m"_3 - "m"_2))]`
APPEARS IN
संबंधित प्रश्न
Which has more heat: 1 g ice at 0℃ or 1g water 0℃? Give reason.
Explain the following temperature vs time graph.

What do you mean by the statement?
'The specific latent heat capacity of fusion of ice is 336 J per g'?
Define the following terms:
(i) Specific latent heat,
(ii) Specific latent heat of fusion.
Explain why water is used in hot water bottles for fomentation and also as a universal coolant.
Explain the meaning of the term latent heat. State its S. I. unit.
The specific latent heat of vaporisation of steam is 2260 J/g. Comment on this.
Explain, why no tracks are left on the ice during ice skating?
Explain the meaning of greenhouse effect.
State two advantages of the high specific latent heat capacity of steam, which is about 226 × 104 J/kg?
When ice is converted into water : constant temperature : : before the water evaporates : _______
Find the odd one out and give its explanation.
Write the name.
The phase in which solid substances are converted into liquid.
600 g of copper at 50°C is mixed with lOOOg water at 20°C. Find the final temperature of the mixture. The specific heat capacity of copper is 0.4 Jg-1°C-1 and that of water is 4.2 Jg-1°C-1
Calculate the amount of heat required to convert 200g of ice at 0°C into the water at 0°C Specific latent heat of fusion of ice = 336 Jg-1
Calculate the total amount of heat energy required to melt 200 g of ice at 0°C to water at 100°C. (Specific latent heat of ice = 336 Jg-1, specific heat capacity of water = 4.2 Jg-1 °C-1)
20 g of ice at 0°C absorbs 10,920 J of heat energy to melt and change to water at 50°C. Calculate the specific latent heat of fusion of ice. Specific heat capacity of water is 4200 J kg-1 K-1.
