Advertisements
Advertisements
प्रश्न
What observation you will record and how will you determine the specific latent heat of fusion of ice?
उत्तर
A clean and dry copper calorimeter with a strirrer, is weighed when empty, again weighed when filled nearly 2/3 with water. Then initial temperature is noted. Some pieces of dry ice are transferred into the calorimeter. The calorimeter is placed inside the box and is continuously stirred till the whole of ice is melted. The final temperature is carefully noted. Then the mass of calorimeter with water and ice is again weighed.
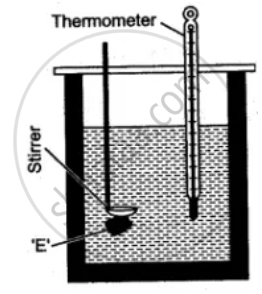
The observations recorded are:
Mass of empty calorimeter with stirrer = m1 gm.
Mass of calorimeter + water = m2 gm.
Initial temperature of water = t1 °C
Final temperature of mixture = t2 °C
Mass of calorimeter + Water + Ice (in the form of water) = m3 gm.
Temperature of ice = 0 °C
If L j/gm be the specific latent heat of fusion of ice and sc be the specific heat capacity of copper, then:
Mass of ice method = (m3 - m1) gm
Heat required by ice water at 0 °C to rise to t2 °C
= (m3 - m2) × 4.2 × (t2 - 0)
Heat lost by calorimeter = m1 × Sc × (t1 - t2)
Heat lost by water = (m2 - m1) × 4.2 × (t1 - t2)
From the heat equation,
Heat gained = Heat lost
(m3 - m2) L + (m3 - m2) × 4.2 × t2 = m1 × sc (t1 - t2) + (m2 - m1) × 4.2 × (t1 - t2)
Therefore,
L = `[(("m"_1"s"_"c")("t"_1 - "t"_2) + ("m"_2 - "m"_1) xx 4.2 xx ("t"_1 - "t"_2) - ("m"_3 - "m"_2) xx 4.2 xx "t"_2)/(("m"_3 - "m"_2))]`
APPEARS IN
संबंधित प्रश्न
State two factors upon which the rate of emission of thermions depends.
State the effect of an increase of impurities on the melting point of ice.
What is the energy absorbed during the phase change called?
A refrigerator converts 100g of water at 20℃ to ice at – 10℃ in 73.5 min. Calculate the average rate of heat extraction in watt. The specific heat capacity of water is 4.2 J kg-1 K-1, specific latent heat of ice is 336 J g-1 and the specific heat capacity of ice is 2.1 J kg-1 K-1.
Explain the following temperature vs time graph.

Liquid ammonia is used in ice factory for making ice from water. If water at 20°C is to be converted into 2 kg ice at 0°C, how many grams of ammonia are to be evaporated? (Given: The latent heat of vaporization of ammonia = 341 cal/g)
Explain the following temperature vs time graph.

When 1 g of ice at 0 °C melts to form 1 g of water at 0 °C then, is the latent heat absorbed by the ice or given out by it?
Why water get cooled in a ‘Surahi’ in hot season?
Define the term ‘specific latent heat of fusion’ of a substance.
Explain the statement; “The specific latent heat of vaporization of wafer is 2260 × 103 J/kg”.
Calculate the total amount of heat required to convert 100g ice at 0°C to steam at 100°C.
(Specific latent heat of fusion of ice = 336 J/g, specific latent heat of vaporization of steam = 2260 J/g, specific heat capacity of water = 4.2 J/g°C).
Find the odd one out and give its explanation.
Define specific latent heat capacity
20 g of ice at 0°C absorbs 10,920 J of heat energy to melt and change to water at 50°C. Calculate the specific latent heat of fusion of ice. Specific heat capacity of water is 4200 J kg-1 K-1.
