Advertisements
Advertisements
प्रश्न
Which metal in the first series of transition metals exhibits +1 oxidation state most frequently and why?
उत्तर
Cu (3d10 4s1) usually shows a +1 oxidation state and forms Cu+ ion (3d10), which has a more stable configuration.
APPEARS IN
संबंधित प्रश्न
Complete the following chemical equations:
`(i) Cr_2O_7^(2-)+6Fe^(2+)+14H^+ ->`
`(ii) 2CrO_4^(2-)+2H^+ ->`
`(iii) 2MnO_4^-+5C_2O_4^(2-)+16H^+ ->`
In 3d series (Sc to Zn), which element has the lowest enthalpy of atomisation and why?
Out of Mn3+ and Cr3+, which is more paramagnetic and why ?
(Atomic nos. : Mn = 25, Cr = 24)
Which is a stronger reducing agent, Cr2+ or Fe2+ and why?
Calculate the ‘spin only’ magnetic moment of \[\ce{M^{2+}_{ (aq)}}\] ion (Z = 27).
How is the variability in oxidation states of transition metals different from that of the non-transition metals? Illustrate with examples.
Describe the oxidising action of potassium dichromate and write the ionic equation for its reaction with iodide.
Give reasons Iron has the higher enthalpy of atomization than that of copper.
Give reasons: Sc3+ is colourless in aqueous solution whereas Ti3+ is coloured.
How is potassium dichromate prepared from chrome iron ore?
Maximum magnetic moment is shown by ____________.
When a brown compound of manganese (A) is treated with \[\ce{HCl}\] it gives a gas (B). The gas taken in excess, reacts with \[\ce{NH3}\] to give an explosive compound (C). Identify compounds A, B and C.
Identify the metal and justify your answer.
\[\ce{MO3F}\]
Transition metals can act as catalysts because these can change their oxidation state. How does \[\ce{Fe(III)}\] catalyse the reaction between iodide and persulphate ions?
The orientation of an atomic orbital is governed by
Which of the following transition metal is not coloured?
The electrode potential of M2+/M of 3d-series elements shows the positive value for ______.
Which one among the following metals of the 3d series has the lowest melting point?
The trend of which property is represented by the following graph?
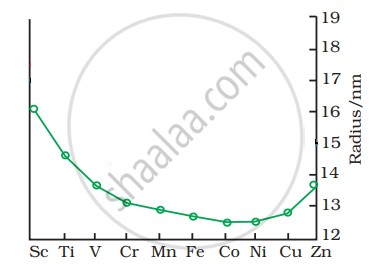
Explain the magnetic properties of d-block (or transition) elements.
