Advertisements
Advertisements
प्रश्न
Why is Butan-1-ol optically inactive but Butan-2-ol is optically active?
उत्तर
Butan-2-ol has a chiral centre, that is, butan-2-ol has a carbon atom bonded to four different substituents.
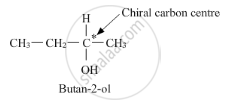
Thus, it is optically active. On the other hand, butan-1-ol does not have any chiral carbon atoms.
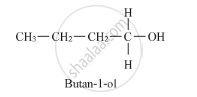
Therefore, it is optically inactive.
APPEARS IN
संबंधित प्रश्न
Draw the geometrical isomers of complex [Pt(NH3)2Cl2].
Indicate the types of isomerism exhibited by the following complex and draw the structures for these isomers:
K[Cr(H2O)2(C2O4)2]
How many geometrical isomers are possible in the following coordination entity?
[Cr(C2O4)3]3−
Draw the structure of optical isomers of [PtCl2(en)2]2+.
Draw all the isomers (geometrical and optical) of [Co(NH3)2Cl2(en)]+.
Draw the geometrical isomers of complex \[\ce{[Pt(en)2Cl2]^2+}\].
Name the type of isomerism shown by the following pair of compounds:
[CoCl(H2O)(NH3)4]Cl2 and [CoCl2(NH3)4]Cl.H2O
The complex [(Pt(Py)(NH3)BrCl] will have how many geometrical isomers?
Indicate the type of isomerism exhibited by the following complex and draw the structure for this isomer: \[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
