Advertisements
Advertisements
प्रश्न
Write chemical equation for the event.
Electrolysis of alumina is done.
उत्तर १
The electrolysis of alumina is carried out in a steel tank lined inside with graphite. The graphite lining serves as cathode. Anode is also made up of graphite rods hanging in the molten mass. The electrolyte consists of alumina dissolved in fused Cryolite(Na3AlF6) and Fluorspar(CaF2). Cryolite lowers the melting point of alumina and fluorspar increases the fluidity of the mass so that the liberated aluminum metal may sink at the bottom of the cell. When electric current is passed through this mixture, the aluminum is collected at the cathode in molten state and sinks at the bottom.

Ionization of Alumina:2Al2O3 → 6O-2 + 4Al+3
Reaction at Cathode: 4Al+3 + 12e- → 4Al
Reaction at Anode: 6O-2 → 3O2 + 12e-, C + O2 → CO2
उत्तर २
When electrolysis of alumina is done, aluminium is formed at the cathode and oxygen gas liberated at the anode.
Cathode:
\[\ce{Al^{3+} + 3e^- -> Al_{(l)}}\] (Reduction)
Anode:
\[\ce{2O^2- -> O_{2(g)} + 4e^-}\] (Oxidation)
संबंधित प्रश्न
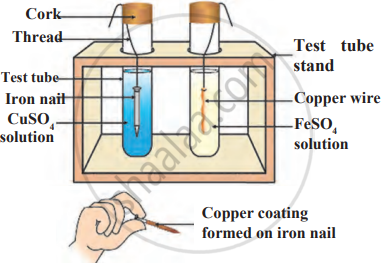
What do you observe when a few pieces of iron are dropped in a blue solution of copper sulphate?
What do you observe when silver nitrate is added to a solution of sodium chloride?
What do you observe when ferrous sulphate solution is added to an aqueous solution of sodium hydroxide.
Fill in the blank
When a piece of copper is added to silver nitrate solution, it turns ............in colour.
How will you obtain Magnesium oxide from magnesium.
Also give balanced equations for the reactions
How will you obtain Zinc chloride from zinc.
Also give balanced equations for the reactions
What do you observe when Magnesium ribbon is burnt in oxygen.
Write chemical equation for the event.
Iron filings are dropped in aqueous solution of copper sulphate.
Write a chemical equation for the following event.
Zinc oxide is dissolved in dilute hydrochloric acid.
Divide the metals Cu, Zn, Ca, Mg, Fe, Na, Li into three groups, namely reactive metals, moderately reactive metals and less reactive metals.
Explain the following reaction with the balanced equation.
Sodium burns in air
Explain the following reaction with the balanced equation.
Reaction of aluminium with oxygen
Explain the following reaction with the balanced equation.
Sulphur burns in air
Observe the following diagram and identify the type of reaction and write observation.
Which among the following statements is incorrect for magnesium metal?
Arrange the following as per the instruction given in the bracket:
Al, K, Mg, Ca (decreasing order of its reactivity)
