Advertisements
Advertisements
प्रश्न
Write the molecular formula and structure of benzene.
उत्तर
The molecular formula of benzene is C6H6.
Its structural formula is :
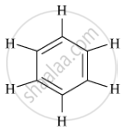
APPEARS IN
संबंधित प्रश्न
Write the name and general formula of a chain of hydrocarbons in which an addition reaction with hydrogen is possible. State the essential condition for an addition reaction. Stating this condition, write a chemical equation giving the name of the reactant and the product of the reaction.
List two reasons for carbon forming a large number of compounds. Name the type of bonding found in most of its compounds. Why does carbon form compounds mainly by this kind of bonding?
What is meant by isomers?
Draw the structures of possible isomers of butane, C4H10.
You are given the following molecular formulae of some hydrocarbons:
C5H8; C7H14; C6H6; C5H10; C7H12; C6H12
Which three formulae represent open chain unsaturated hydrocarbons having double bonds?
Name the functional group which always occurs in the middle of a carbon chain.
Pentane has the molecular formula C5H12. It has ______.
Hydrocarbons are mainly composed of which of these?
The number of isomers of pentane is ______.
How will you prove that C4H8 and C5H10 are homologues?
